
Concept explainers
The predictions that conclude that radioactive emission consists of positively charged particles, negatively charged particles, and neutral particles.
Answer to Problem 1RQ
Solution:
Testing the deflection of
Explanation of Solution
Introduction:
Rutherford, in 1899,
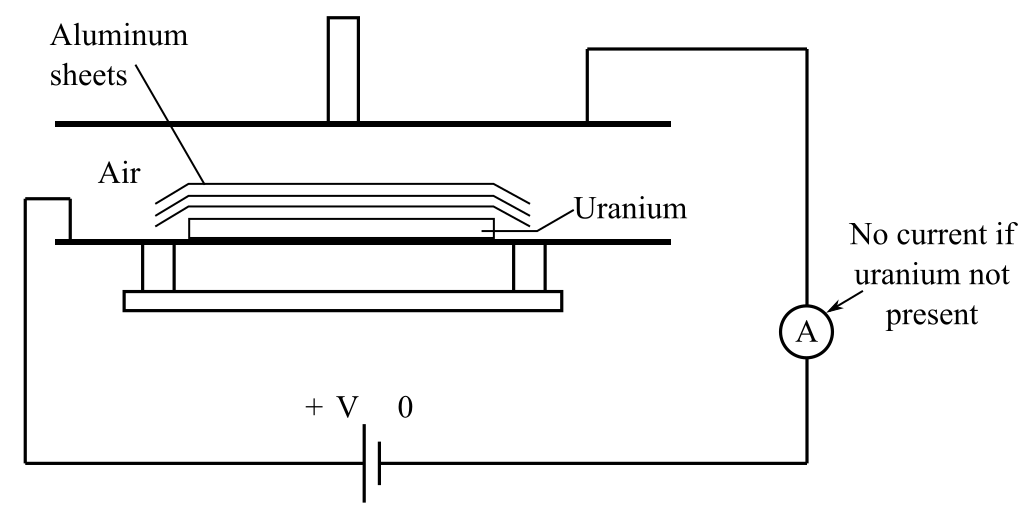
Explanation:
By passing radiation through a magnetic field, the behavior of particles in radiation can be observed.
Rutherford conducted an experiment where the radiation emitting from a radioactive sample passes through a 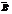 field pointing into the page, and allowed them to fall on a scintillation screen placed perpendicular to the incident beam.
field pointing into the page, and allowed them to fall on a scintillation screen placed perpendicular to the incident beam.
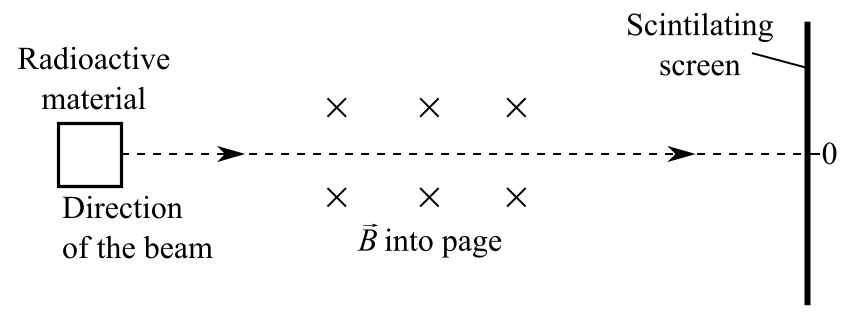
By the right-hand rule, if radiation contains positively charged particles, then the particle should deflect upwards due to the magnetic field. And, if radiation contains negatively charged particles, it should deflect downwards on the scintillation screen.
The outcome was that the scintillation screen glowed in three places: straight ahead, deflected up, and deflected down. And, the downward deflection was much farther than the upward deflection from the un-deflected radiation.
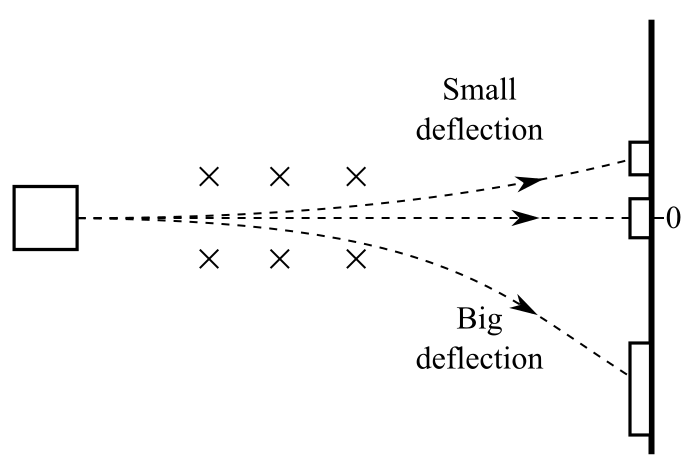
Conclusion:
So, the experiment suggested that the radioactive radiation consists of three components: positively charged particles (alpha particles), negatively charged particles (beta particles), and neutral particles (gamma rays) that were not deflected by the magnetic field.
Want to see more full solutions like this?
Chapter 29 Solutions
College Physics
- An electron can interact with a nucleus through the beta-decay process: ZAX+eY+ve Write the complete reaction equation for electron capture by 7Be . Calculate the energy released.arrow_forwardData from the appendices and the periodic table may be needed for these problems. 53. You can sometimes find deep red crystal vases in antique stores, called uranium glass because their color was produced by doping the glass with uranium. Look up the natural isotopes of uranium and their halflives, and calculate the activity of such a vase assuming it has 2.00 g of uranium in it. Neglect the activity of any daughter nuclides.arrow_forwardA piece of wood from an ancient Egyptian tomb is tested for its carbon-14 activity. It is found to have an activity per gram of carbon of A = l0 decay/min.g. What is the age of the wood?arrow_forward
 University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
 An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning





