
Concept explainers
a)
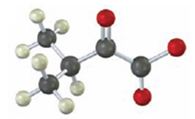
Interpretation:
To identify the amino acid that is a catabolic precursor of the given α-keto acids:
Answer to Problem 17VC
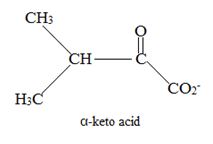
Explanation of Solution
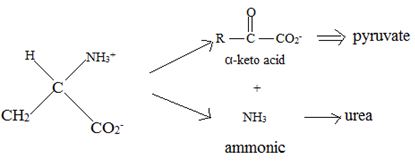
Catabolism of proteins occurs by a discrimination of α – amino acids according to the following reaction:
The retro symmetric strategy is used to derive synthons and starting materials.
b)
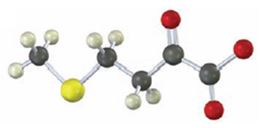
Interpretation:
To identify the amino acid that is a catabolic precursor of the given α-keto acids:
Answer to Problem 17VC
The corresponding α amino acid precursor is respectively:
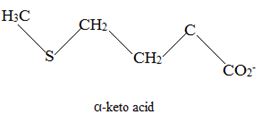
Explanation of Solution
Catabolism of proteins occurs by a discrimination of α – amino acids according to the following reaction:
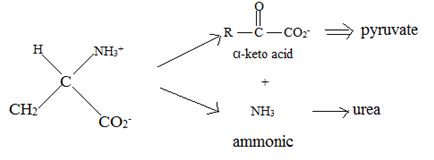
Thus, the α – keto acid derives its structure from its precursor parent amino acid by replacing the 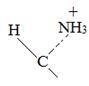 segment by
segment by 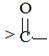 .
.
This proves the above assigned structures for the α – amino acids.
The retro symmetric strategy is used to derive synthons and starting materials.
Want to see more full solutions like this?
Chapter 29 Solutions
ORGANIC CHEMISTRY(LL)+OWLV2 4 TERM>BI<
- Draw the structure of the -keto acid produced from the oxidative deamination of each of the following amino acids. a. Glutamate b. Cysteine c. Alanine d. Phenylalaninearrow_forwardDraw the structure of the -keto acid produced from the oxidative deamination of each of the following amino acids. a. Glycine b. Leucine c. Aspartate d. Tyrosinearrow_forwarddraw the chemical structures of the reactant and product of the following reactions: a.) addition of a phosphate group to (ex: phosphorylation of) glycose b.) addition of a phosphate group to (ex: phosphorylation of) fructose-6-phosphate c.) addition of a phosphate group to (ex: phosphorylation of) phosphoenolpyruvatearrow_forward
- Suggest a name for an enzyme that catalyzes each of the following reactions. a. Hydrolysis of lactose b. Oxidation of nitrite c. Decarboxylation of citrate d. Reduction of oxalatearrow_forwardWhat type of specificity (absolute, group, linkage, or stereochemical) is associated with each of the following enzymes? a. A deaminase b. A phosphatase c. Maltase d. L-Lactate dehydrogenasearrow_forwardCells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism. a. What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination? b. What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?arrow_forward
- Define the following terms:a. tautomerizationb. tautomerc. amphibolic pathwayd. electron transport systeme. decarboxylation reactionarrow_forwardTriose phosphate isomerase catalyzes the conversion of dihydroxyacetone phosphate (DHAP) into glyceraldehyde-3-phosphate (G3P). Which of the following illustrates this chemical reaction? a) A b) B c) C d) Darrow_forwardHow does the direct oxidative deamination of L-amino acids different from glutamate occur? 1) the oxidases act together with FMN 2) the oxidases act together with NAD 3) the released hydrogen enters the respiratory chain 4) the released hydrogen is transferred onto oxygen and hydrogen peroxide is formed 5) the obtained products are biogenic amines a) 2+4 b) 1+3 c) 1+4 d) 2+3 e) 4+5arrow_forward
- Define acetylcholinesterasearrow_forwardDraw the reaction then identify the reaction mechanism that took place, SN1, SN2 and etc. Metabolic pathway: Conversion of Pyruvate to Acetyl CoA Citric acid cycle Gluconeogensis pathway (pyruvate to glucose)arrow_forwardWhat is the structure of the a@keto acid formed from transamination of the following amino acids?(a) Glutamic acid (b) Alaninearrow_forward

 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





