
Concept explainers
Interpretation:
A model of naphthalene is given. The position of multiple bonds in it is to be shown. The number of possible resonance structures for naphthalene is to be stated and their structures are to be drawn.
Concept introduction:
Hydrogen is monovalent. Carbon is tetravalent and it can form four bonds. The position of double bonds can be identified by looking for carbons in the model having only three bonds. Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To determine:
The position of multiple bonds in the given model of naphthalene, C10H8. The number and structures of possible resonance structures for naphthalene are to be drawn
Answer to Problem 20VC
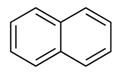
The positions of multiple bonds in naphthalene are shown in the structure given below.
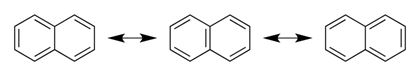
Naphthalene has three resonance forms as shown.
Explanation of Solution
In the model of naphthalene shown all the carbons have formed only three bonds and all the carbons require one more bond to satisfy their fourth valence. This fourth valence of each carbon can be satisfied by placing five double bonds between adjacent carbons as shown.
By shifting the position of double bonds, in total, three resonance structures can be drawn.
The positions of multiple bonds in naphthalene are shown in the structure given below.

Naphthalene has three resonance forms as shown.

Want to see more full solutions like this?
Chapter 2 Solutions
ORGANIC CHEM.,VOL.1-W/ACCESS >CUSTOM<
- Find all molecules/ions with resonance structures and draw them. HCN CH3OH SeF6 NO2- AsF5 XeF2 TeF5- H2CO SF4 XeF4 PO43- BrF3 NH3 CH3NH2arrow_forwardNhy is resonance structure A and resonance structure B not the same structure? If you rotate the molecule like a spicket handle (l. e. clockwise with the C at the center and the R group remaining in place as you look down the C - R bond) wouldn't you get the same thing? Why are these not equivalent structures? Resonance structure A t Resonance structure B Actual structurearrow_forwardWhich of the following species is a valid resonance structure of A? Usecurved arrows to show how A is converted to any valid resonancestructure. When a compound is not a valid resonance structure of A,explain why not.arrow_forward
- #1: What is the formal charge of bromine in the structure of bromate, BrO3-? a) 0 b) +1 c) +2 d) -1 #2: Carbonate (CO32-) has three resonance structures. Choose the stayement that best expains the meaning of those 3 resonance structures. a) Carbonate consists of two single CO bonds and one double CO bond. b) Carbonate consists of one single structure which is an average of the three resonance structures. c) Carbonate quickly flips back and forth between three structures. d) Three different structures for carbonate coexist. #3: When compaing a carbon-carbon double bond (C==C) and a carbon-carbon single bond (C-C), the double bond is a) longer and shorter b) longer and weaker c) shorter and stronger d) shorter and weakerarrow_forward1. Draw Lewis Structures for the following compounds. (Be sure to minimize formal charges and draw resonance structures where appropriate.): XeF2O, ClO4-, HNO2, CH3CH(NH2)COOH (amino acid alanine).arrow_forwardWhich has a greater dipole moment? CH2Cl2 or CH2F2 The book states that a C-Cl bond has a greater dipole moment than a C-F bond, regardless of F's larger electronegativity, because the C-Cl has a larger bond length. Does the same logic apply?arrow_forward
- 1) what is the formal charge of G? a. -1 b. 0 c. +1 2) what is the formal charge of the labelled atom (refer to the blue arrow)? a. -1 b. 0 c. +1arrow_forwardDoes CH3COCH3 has molecular resonance? If so, show all possible forms.arrow_forwardWhich of the following choices depicts the correct resonance arrows to show the conversion between the Q resonance form to the R resonance form?arrow_forward
- Draw the three resonance structures for (a) the cyanate ion (NCO−) and (b) the isocyanate ion (CNO−). In each case, rank the resonance structures in order of increasing importance. Be sure to include all lone pair electrons and nonzero formal charges.a) NCO−arrow_forwardDraw the three non-ionic contributions to the resonance structure of naphthalene, C10H8 .arrow_forward8. Compute for the formal charge (FC) on each atom in the following compounds. a. H3N - BH3 b. [H2CNH2]+ c. H3O+ d. HCO3- 9. Draw Lewis structures for the following compounds, showing an appropriate formal charges. a. [CH3OH2]+ b. [HONH3]+ c. NaBH3CN d. CH3NH3+ 10. For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they would have the same energy. a. [CH3OCH2]+ b. H2SO4 c. CO3-2 d. H2C=CH-+CH2 e. [CH3C(OCH3)2 ]+ 11. Draw the complete Lewis structures for the following condensed structural formulas. a. CH3(CH2)3CH(CH3)2arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





