
Concept explainers
The density of the fluid in the left arm.
Answer to Problem 165P
The density of the fluid in the left arm is
Explanation of Solution
Given information:
The fluid is incompressible and stay separate and do not mix with each other, the angular velocity of rotation of the U-tube is
The figure shows the different pressure points.
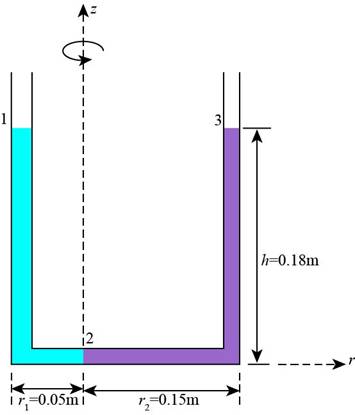
Figure-(1)
Write the expression for converting the angular velocity from
Here, the angular velocity in
Write the expression for the pressure difference for the fluid at point 2.
Here, the pressure of the fluid at point 1 is
Write the expression for the pressure difference for the water at point 3.
Here, the pressure of the water at point 2 is
Write the expression for the different points at atmospheric pressure.
Calculation:
Substitute
Substitute
Substitute
Substitute
Conclusion:
The density of the fluid in the left arm is
Want to see more full solutions like this?
Chapter 3 Solutions
Fluid Mechanics Fundamentals And Applications
- A. Nitrogen at a temperature of 310°F occupies a volume of 10.58 cu. ft. If the initial volume is maintained constant and the pressure tripled, determine the final temperature in K. B. If 100 ft3 of atmospheric air at zero Fahrenheit temperature are compressed to a volume of 1 ft3 at temperature of 200°F, what will be the gauge pressure of the air in psi?arrow_forwardA 10 ft3 vessel of hydrogen at a pressure of 305 psia is vigorously stirred by paddles until the pressure becomes 400 psia. Determine the change in internal energy.arrow_forwardA U-manometer shown in the figure is initially at rest at the current level of mercury. Determine the maximum amount of water that can be added on the right leg of the manometer in grams if h = 40 cm, DA = 2.5 cm, and DB = 5 cm.arrow_forward
- A rigid tank contains 20 lbm of air at a pressure of 20 lbf/in2 and 70°F. More air is added to the tank until the pressure and temperature rise to 35 lbf/in2 and 90°F, respectively. Determine the mass of air added to the tankarrow_forwardThe gage pressure in a liquid at a depth of 2.5 m is read to be 28 kPa. Determine the gage pressure in the same liquid at a depth of 9 m.arrow_forwardDetermine the pressure and specific volume of water at 20 C that has an internal enegy of 1200 KJ/kg.arrow_forward
- Determine the mass of air in lbm when the pressure is 21.83 psi and the temperature is 84.9°F in a closed chamber with dimensions 25.5 ft x 15.6 ft x 19.2 ft. Assume air to be an ideal gas.arrow_forwardA 3 m diameter , 7 m long cylindrical tank is completely filled with water. The tank is pulled by a truck on a level road with 7 m long axis being horizontal. Determine the pressure difference in kPa between the front and back ends of the tank along a horizontal line when the truck accelerates at 3 m/s^2. a.21 b.25 c.23 d.28arrow_forwardThe water in a tank is pressurized by air, and the pressure is measured by a multifluid manometer as shown in Figure below. The tank is located on a mountain at an altitude of 1400 m where the atmospheric pressure is 85.6 kPa. Take the densities of water, oil, and mercury to be 1000 kg/m3, 850 kg/m3 and 13600 kg/m3 respectively. 1. Analyse the change in manometric fluid (oil and mercury) with another fluid on manometric height h2 and h3 for the manometer given in figure above.arrow_forward
- The helium-filled blimp shown in the figure below is used at various athletic events. Determine the number of pounds of helium (lbm) within it if its volume is 68,000 ft3 and the temperature and pressure are 80 °F and 14.2 psia, respectively. The gas constant for helium is 1.242×104 (ft-lbf)/(slug-R).arrow_forwardThe thermodynamic process of a substance when it passes through device changes in a constant pressure process. If the initial and final volumes of the substance are 0.4m3and 0.13m3respectively, what is the magnitude of work is required for this process? Is it done on or done by the system? Draw a schematic diagram,andthen plot the state/s and process, whichever is applicable in a t-sdiagramarrow_forwardThe pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air in the tire before the trip is 25°C, the air temperature after the trip isarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





