
Concept explainers
What two
a. an R-group and a hydroxyl group
b. an N–H group and a carbonyl group
c. an amino group and a hydroxyl group
d. an amino group and a carboxyl group
Introduction:
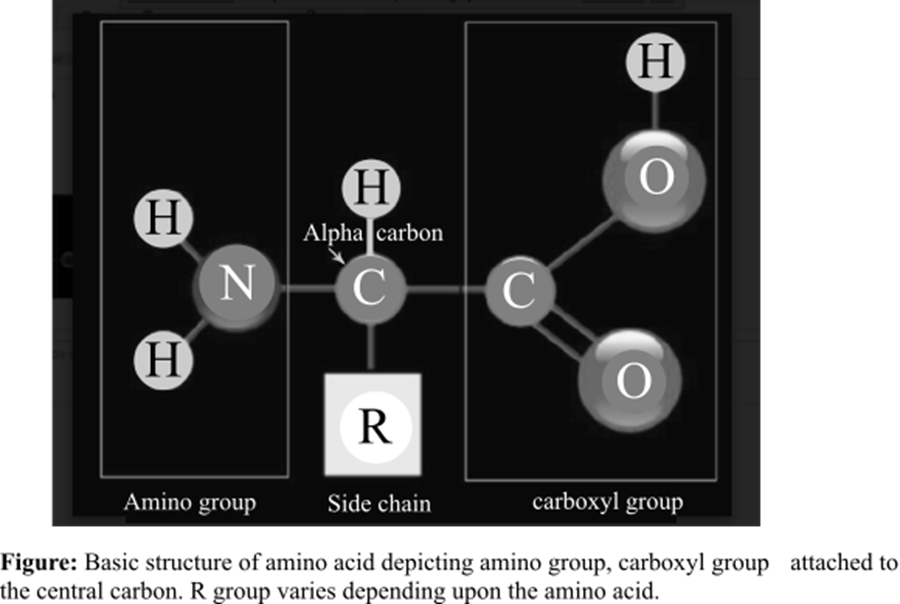
The amino acid is the basic structural unit of the proteins. There are total 20 amino acids found in the living system. At isoelectric point (pH or potential of hydrogen), an amino acid does not have any net charge. The basic structure of amino acids is shown below:
Answer to Problem 1TYK
Correct answer:
An amino group and a carboxyl group
Explanation of Solution
Explanation/Justification for the correct answer:
Option (d) is given as a carboxyl group along with an amino group. Functional groups of amino acids are responsible for bonding between two amino acids. The polypeptide chain consists of several amino acids. When a peptide bond is formed, a hydroxyl (–OH) group is lost from the carboxyl group of an amino acid and an H (hydrogen atom) from the amino group of another amino acid is also lost. This dehydration (loss of one H2O molecule) reaction results in a peptide bond. Hence, option (d) is correct.
Explanation for incorrect answers:
Option (a) is given as an R- group and a hydroxyl group. R group or side chain decides the identity of an amino acid, for example, glycine, which is the simplest amino acid, has a hydrogen atom as its R group. So, it is a wrong answer.
Option (b) is given as an N-H group and a carbonyl group. Any functional group, which has an O (oxygen) atom attached to the C (carbon) atom through double bond (-C=O) is called carbonyl group. COOH (carboxylic acid) is also a type of the carbonyl group. So, it is a wrong answer.
Option (c) is given as an amino group and a hydroxyl group. The carboxylic acid of the amino acid contains one hydroxyl group, which gets lost when the amino acid undergoes peptide bond formation with an amino group of another amino acid. So, it is a wrong answer.
Hence, options (a), (b), and (c) are incorrect.
The amino group and a carboxyl group are functional groups bounded to the central carbon of every free amino acid monomer.
Want to see more full solutions like this?
Chapter 3 Solutions
Biological Science (6th Edition)
- a.Describe the bonds which hold a quaternary protein molecule together. b. Discuss the reasons why glycine and proline are not usually found in an alpha helix of proteins.arrow_forwardConstruct a Venn diagram using the one-letter symbol of the amino acids. Provide a reason that indicates why the amino acids are groups according to the venn diagram.arrow_forwardWrite an essay on the structure and properties of the amino acid L-histidine . (200 Words)arrow_forward
- How many amino acids and peptide bonds does a tetrapeptide have? A) four amino acids and three peptide bonds B) four amino acids and four peptide bonds C) three amino acids and four peptide bonds D) four amino acids and two peptide bondsarrow_forwardWhich functional groups are present in digitoxin? a. To what lipid family does the complex ring drawn to the right of the structure belong? b. Identify the type of glycosidic bond that joins each of the monosaccharide residue in the molecule c. Draw the open form of the monosaccharide used to produce the trisaccharide portion of digitoxin.arrow_forwardBetween which two amino acids do disulfide bonds form? A.Alanine-Glycine B.Serine-Threonine C.Cysteine-cysteine D.Methionine-methioninearrow_forward
- Which bond in a polypeptide backbone does not freely rotate? a. The bond between CO and NH b. The bond between C-alpha and NH c. The bond between C-alpha and CO d. The bond between C-alpha and C-beta c. d.arrow_forwardWrite down the reactions of serine and arginine amino acids with carboxyl and amino groups.arrow_forwardWhich statement describes the bases pairing in nucleic acids? Purine bases always pair with other purine bases Purine bases can only pair with pyrimidine bases Adenine cannot pair with either uracil or thymine Hydrogen bonding can only occur between the pyrimidine bases Guanine is always paired with adeninearrow_forward
- Name and draw the structures of the amino acids that fit the following descriptions:(a) Contains an isopropyl group(b) Contains a secondary alcohol grouparrow_forwardGiven below is the structure of tallose.Answer the following questiona. what is the maximum number of stereoisomers can tallose have?b. how many chiral carbons does it have?c. what is the configuration of tallose is it D or L sugar?d. what type of monosaccharide is it?arrow_forwardBased on what you know about glycosidic bonds, propose a scheme for formation of covalent bonds between the carbohydrate and protein portions of glycoproteinsarrow_forward

 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College


