
In 1976, a team of chemists in the United Kingdom was developing new insecticides by modifying sugars with chlorine (Cl2), phosgene (Cl2CO), and other toxic gases. One young Member of the team misunderstood his verbal instructions to “test” a newly made candidate substance. He thought he had been told to “taste” it. Luckily for him, the substance was not toxic, but it was very sweet. It became the food additive sucralose.
Sucralose has three chlorine atoms substituted for three hydroxyl groups of sucrose (table sugar). It binds so strongly to the sweet-taste receptors on the tongue that the human brain perceives it as 600 times sweeter than sucrose. Sucralose was originally marketed as an artificial sweetener called Splenda”, but it is now available under several other brand names.
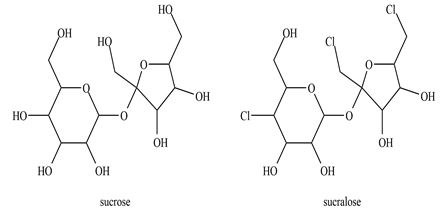
Researchers investigated whether the body recognizes sucralose as a carbohydrate. They began by feeding sucralose labeled with 14C to volunteers. Analysis of the radioactive molecules in the volunteers’ urine and feces showed that 92.8 percent of the sucralose passed through the body without being altered. Many people are worried that the chlorine atoms impart toxicity to sucralose. How would you respond to that concern?
Trending nowThis is a popular solution!

Chapter 3 Solutions
BIOLOGY:CONCEPTS+APPL.(LOOSELEAF)
- What color of light results from the addition of cyan light and red light? green blue white magentaarrow_forwardA food factory wants to test the film strength of different types of starch, but the film strength is affected by the film thickness, and it is difficult to control the film thickness during the production process. Only the thickness data of various film samples can be recorded, but when the manufacturer wants to test the film strength , the influence of film thickness can be excluded. How should the manufacturer design the experiment? In order to compare the strength of the film and exclude the influence of the thickness of the film, how should the experiment be randomization?arrow_forwardWhich of the following types of photons (packets of pure electromagnetic energy) have the highest energy level? visible light infrared microwaves ultra-violetarrow_forward
- Which of the following wavelengths in the electromagnetic spectrum has the most energy? a. microwaves b. visible light c. ultraviolet d. radio wavesarrow_forwardIf a bottle of aspirin tablets has the aroma of vinegar, it is time to discard those tablets. Explain why, and include a chemical equation in the Explanation.arrow_forwardWhat color does an object appear if it absorbs light at both 420-500 nm and at 620-700 nm? a. Red b. Black c. Purple d. Green e. Deep bluearrow_forward
- Name the colors in a color spectrum of visible light from low to high wavelength.arrow_forwardA multinational company outside india tried to sell new varieties of turmeric without proper patent rights.what is such an act referred to?arrow_forwardAnthocyanins are another type of plant pigment that are purple or red. Say you run a chromatography experiment using the same setup as you did in this lab, and you find that anthocyanins do not move from the line of origin. Assuming the solvent traveled the same distance as in your experiment (D in Table 1 above), what is the Rf value of anthocyanin?arrow_forward
- Imagine you have an oil/water mixture where the oil is non-polar and the water is polar. Your friend grabs table salt (sodium chloride) and throws it into the mixture. What do you expect to see? If your friend has no chemistry background, what would you say to your friend to explain the result?arrow_forwardWhat features of the scent molecules of apples and oranges determine their volatility and odors?arrow_forwardSuppose you conduct direct visible spectrophotometry with red fruit punch as your solution. Which wavelengths of light would you expect to be strongly absorbed? Which wavelengths are not strongly absorbed? Explain.arrow_forward

 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning



