
Campbell Biology in Focus (2nd Edition)
2nd Edition
ISBN: 9780321962751
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 2TYU
Which functional group is not present in this molecule?
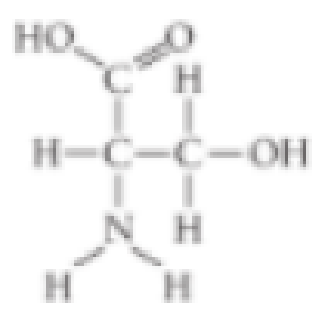
- A. carboxyl
- B. sulfhydryl
- C. hydroxyl
- D. amino
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Answer (a.) If compound X is aspartic acid and compound Y is lysine
Speculate on the properties of proteins and peptides if none of the common amino acids contained sulfur.
Which is not true of RNA?
a. It is usually single-stranded.
b. It functions in the cytoplasm.
c. It contains uracil.
d. It contains ribose.
e. There are 4 functional types.
Chapter 3 Solutions
Campbell Biology in Focus (2nd Edition)
Ch. 3.1 - How are gasoline and fat chemically similar?Ch. 3.1 - Which molecules in Figure 3.4a re isomers? For...Ch. 3.1 - Prob. 3CCCh. 3.1 - Prob. 4CCCh. 3.2 - How many molecules of water are needed to...Ch. 3.2 - WHAT IF? Suppose you eat a serving of fish. What...Ch. 3.3 - Write the formula for a monosaccharide that has...Ch. 3.3 - A dehydration reaction joins two glucose molecules...Ch. 3.3 - WHAT IF? After a cow is given antibiotics to treat...Ch. 3.4 - Compare the structure of a fat (triglyceride) with...
Ch. 3.4 - Why are human sex hormones considered lipids?Ch. 3.4 - Prob. 3CCCh. 3.5 - Why does a denatured protein no longer function...Ch. 3.5 - What parts of a polypeptide participate in the...Ch. 3.5 - WHAT IF? Where would you expect a polypeptide...Ch. 3.6 - DRAW IT Go to Figure 3.27a and, for the top three...Ch. 3.6 - Prob. 2CCCh. 3.7 - How would sequencing the entire genome of an...Ch. 3.7 - Given the function of DNA, why would you expect...Ch. 3 - Prob. 1TYUCh. 3 - Which functional group is not present in this...Ch. 3 - MAKE CONNECTIONS Which chemical group is most...Ch. 3 - Prob. 4TYUCh. 3 - Which of the following statements concerning...Ch. 3 - The structural level of a protein least a fleeted...Ch. 3 - Enzymes that break down DNA catalyze the...Ch. 3 - Prob. 8TYUCh. 3 - The molecular formula for glucose is C6H12O6. What...Ch. 3 - Construct a table that organizes the following...Ch. 3 - Prob. 11TYUCh. 3 - Prob. 12TYUCh. 3 - FOCUS ON ORGANIZATION Proteins, which have diverse...Ch. 3 - Prob. 14TYUCh. 3 - SYNTHESIZE YOUR KNOWLEDGE Given that the function...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (10th Edition)
Why are mutants used as test organisms in the Ames test?
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Some people consider Pasteur or Koch to be the Father of Microbiology, rather than Leeuwenhoek. Why might they ...
Microbiology with Diseases by Body System (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Deduce the number of isomers of fructosearrow_forwardWhich is true about carbon atoms? a. Each of the orbitals in the second shell has one electron in it. b. Only the 2p orbitals have unpaired electrons c. Carbon has high electronegativity d. Carbon binds hydrogen with polar covalent bondsarrow_forward(a) What type of linkage is present in disaccharides?(b) Write one source and deficiency disease of vitamin B12.(c) Write the difference between DNA and RNA.arrow_forward
- What was the purpose of using a high temperature and low pH in starch hydrolysis? a. Increase yield b. To increase the pressure c. Increase the rate of reaction d. Decrease the yieldarrow_forwardWhich statement describes the bases pairing in nucleic acids? Purine bases always pair with other purine bases Purine bases can only pair with pyrimidine bases Adenine cannot pair with either uracil or thymine Hydrogen bonding can only occur between the pyrimidine bases Guanine is always paired with adeninearrow_forwardWhich bond in a polypeptide backbone does not freely rotate? a. The bond between CO and NH b. The bond between C-alpha and NH c. The bond between C-alpha and CO d. The bond between C-alpha and C-beta c. d.arrow_forward
- Give a clear handwritten answer of this in which who have long chain of atoms in which molecules contain given below..?Explain.. (A) Lipids (B) DNA (C) Proteins (D) Amylosearrow_forwardRead the question carefully & encircle the letter of thecorrect answer.1. Amino acids are the building blocks of which group of biomolecules?a. Proteins b. carbohydrates c. lipids d. nucleic acidsarrow_forwardPhospholipase responsible for the tissue damage after spider bite? a.A1 b.A2 c.D d.Carrow_forward
- Which compound is inorganicarrow_forwardUse the drawing to answer the following: a. Label the bases as A, c, G, or T b. Draw in the Hydrogen bonds c. Circle one Glycosidic bond d. Star one Phosphodiester linkagearrow_forwardWhat is the reaction of fats with strong bases producing salts of the fatty acids known as metallic esters and glycerol? a. acid hydrolysis b. alkaline hydrolysis c. water hydrolysis d. oxidationarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license