
Concept explainers
(a)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are sixteen
Explanation of Solution
The line drawing of the given molecule is:
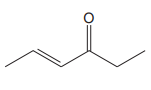
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
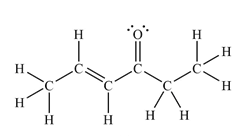
This shows fourteen single bonds, two double bonds, and two lone pairs. Therefore, the molecule contains a total of sixteen
A single bond between two atoms is a
(b)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are sixteen
Explanation of Solution
The line drawing of the given molecule is:
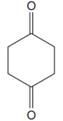
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
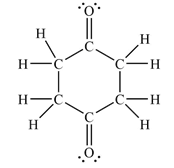
This shows fourteen single bonds, two double bonds, and four lone pairs. Therefore, the molecule contains a total of sixteen
A single bond between two atoms is a
(c)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are twelve
Explanation of Solution
The line drawing of the given molecule is:
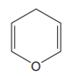
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
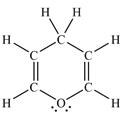
This shows twelve single bonds, two double bonds, and two lone pairs. Therefore, the molecule contains a total of twelve
A single bond between two atoms is a
(d)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are nineteen
Explanation of Solution
The line drawing of the given molecule is:
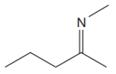
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
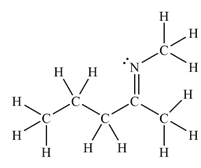
This shows eighteen single bonds, one double bond and one lone pair. Therefore, the molecule contains a total of nineteen
A single bond between two atoms is a
(e)
Interpretation:
The number of
Concept introduction:
In order to determine the number of
In the Lewis structure, a single bond represents an electron pair in a
Answer to Problem 3.18P
There are eleven
Explanation of Solution
The line drawing of the given molecule is:
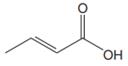
This must be converted to a Lewis structure showing all atoms and lone pairs before the number of bonds of different types and the electrons in nonbonding MOs can be counted.
The Lewis structure of the molecule showing all atoms, bonds and lone pairs can be drawn as:
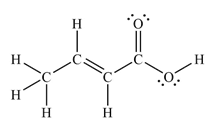
This shows nine single bonds, two double bonds, and four lone pairs. Therefore, the molecule contains a total of eleven
A single bond between two atoms is a
Want to see more full solutions like this?
Chapter 3 Solutions
ORG. CHEM. LL W/SSM+SG+ACCESS
- For problem 3, why does N keep its electrons in addition to taking the H+ from the other molecule? Didn't it give its electrons to the other molecule?arrow_forwardKindly explain why COSe is the answer in detail.arrow_forwardUse curved arrow notation to show how the rst resonance structure can be converted to the second.arrow_forward
- For the same molecule, which of the images corresponds to the correct Newman projection along the indicated bond? Need help understaniding how to answer question?arrow_forwardImage below, Why is this not a constructual isomer?arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.Zarrow_forward
- match the following to its compound name. *First row only from upper to bottomarrow_forwardProblem What amount (mol) of each ion is in each solution?(a) 5.0 mol of ammonium sulfate dissolved in water(b) 78.5 g of cesium bromide dissolved in water(c) 7.42×1022 formula units of copper(II) nitrate dissolved in water(d) 35 mL of 0.84 M zinc chloridePlan We write an equation that shows 1 mol of compound dissociating into ions. (a) We multiply the number of moles of ions by 5.0. (b) We first convert grams to moles. (c) We first convert formula units to moles. (d) We first convert molarity and volume to moles.arrow_forwardwe can say that the next pair of compounds are:arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
