
Organic Chemistry (Looseleaf)
4th Edition
ISBN: 9780077640194
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3, Problem 3.36P
Explain the observed trend in the melting points for four isomers of molecular formula C7H16.
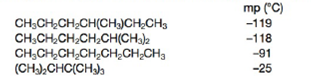
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
c) Draw one possible structure with molecular formula C6H12 that has two tertiary carbon.d) Arrange the following compounds in increasing order of melting point.Hexane, 3-methylhexane, 3-ethylhexane, 2,3-dimethylpentane
List the names of all possible isomers with the molecular formula C5H12. (Tip: It may be helpful to draw all possible isomers on a piece of paper and then name them.)
Draw the two structure isomers for molecules having the molecular formula C4H10
Chapter 3 Solutions
Organic Chemistry (Looseleaf)
Ch. 3 - Prob. 3.1PCh. 3 - Prob. 3.2PCh. 3 - Draw the structure of a compound fitting each...Ch. 3 - Draw structures that fit each description and name...Ch. 3 - What types of intermolecular forces are present in...Ch. 3 - Prob. 3.6PCh. 3 - Explain why the boiling point of propanamide, is...Ch. 3 - Predict which compound in each pair has the higher...Ch. 3 - Prob. 3.9PCh. 3 - Prob. 3.10P
Ch. 3 - a Label the hydrophobic and hydrophilic portions...Ch. 3 - Prob. 3.12PCh. 3 - Prob. 3.13PCh. 3 - Prob. 3.14PCh. 3 - Prob. 3.15PCh. 3 - Nonactin and valinomycin each contain only two...Ch. 3 - Prob. 3.17PCh. 3 - Problem 3.26 Label the electrophilic and...Ch. 3 - Problem 3.27 Considering only electron density,...Ch. 3 - The fact that sweet-tasting carbohydrates like...Ch. 3 - 3.29
Identify the functional groups in the...Ch. 3 - Prob. 3.22PCh. 3 - 3.32 Identify the functional groups in each...Ch. 3 - Draw the seven constitutional isomers having...Ch. 3 - 3.33 Identify each functional group located in the...Ch. 3 - Draw seven constitutional isomers with molecular...Ch. 3 - Prob. 3.27PCh. 3 - Prob. 3.28PCh. 3 - Prob. 3.29PCh. 3 - Intramolecular force of attraction are often...Ch. 3 - 3.40 (a) Draw four compounds with molecular...Ch. 3 - Prob. 3.32PCh. 3 - Explain why CH3CH2NHCH3 has higher boiling point...Ch. 3 - Prob. 3.34PCh. 3 - Prob. 3.35PCh. 3 - Explain the observed trend in the melting points...Ch. 3 - Prob. 3.37PCh. 3 - Prob. 3.38PCh. 3 - Prob. 3.39PCh. 3 - 3.48 Explain why diethylether and have similar...Ch. 3 - Prob. 3.41PCh. 3 - 3.50 Predict the solubility of each of the...Ch. 3 - Prob. 3.43PCh. 3 - Prob. 3.44PCh. 3 - THC is the active component in marijuana, and...Ch. 3 - Prob. 3.46PCh. 3 - Prob. 3.47PCh. 3 - Prob. 3.48PCh. 3 - Label the electrophilic and nucleophilic sites in...Ch. 3 - By using only electron density arguments,...Ch. 3 - Prob. 3.51PCh. 3 - Prob. 3.52PCh. 3 - Prob. 3.53PCh. 3 - Prob. 3.54PCh. 3 - Prob. 3.55PCh. 3 - Recall from section 1.10B that there is restricted...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structural formula(s) for the branched constitutional isomer(s) with the molecular formula C7H16 that have three methyl groups.arrow_forwardWhich one of the following substances would be the most soluble in cyclohexane, C6H12? CH3CH2CH2OH C4H10 NH3 Li2O HClarrow_forwardDraw the structural formula(s) for the constitutional isomer(s) with the molecular formula C7H16 that have an unbranched chain.arrow_forward
- Draw a complete structure for a molecule with the molecular formula C3H8Oarrow_forwardA compound with the molecular formula C6H8N2arrow_forwardThe melting points and boiling points of two isomeric alkanes are asfollows: CH3(CH2)6CH3, mp = −57 °C and bp = 126 °C; (CH3)3CC(CH3)3,mp = 102 °C and bp = 106 °C. Explain why there is a small difference in the boiling points of the two compounds, but a huge difference in their melting points.arrow_forward
- Pentane has a boiling point of 36.1 C, while 1- butanol has a similar mass and has a boiling point of 117.7 C. Explain the difference, including line angle structure of each compound to show the IMFs. Please use two molecules to show IMFs.arrow_forwardCompound: 1-(isopropyl)-2-methylcyclohexane Sketch the line drawing of all the isomers of this compound (showing the 3D nature of the compound with wedges and dashes)arrow_forwardExplain the two acceptable arrangements of atoms for the molecular formula C2H6O ?arrow_forward
- How many possible isomers are there for C2H4Cl2arrow_forwardDraw structural formulas for all constitutional isomers that have the given molecular formula: a) C2H6O and b) C3H7Narrow_forwardC2H6 Draw the structures of the two isomers, known as dimethyl ether and ethanol, of this molecular formula. Label them correctly.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY