
ORGANIC CHEMISTRY (LOOSELEAF)
6th Edition
ISBN: 9781260475630
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3, Problem 36P
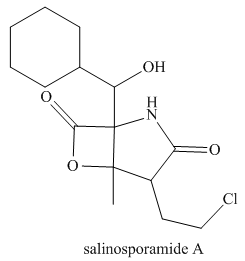
(a) Identify the
from marine sediment. (b) Classify each alcohol, alkyl amide, and amide as
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compounds that contain an N-H group associate by hydrogen bonding.
(a) Do you expect this association to be stronger or weaker than that of compounds containing an O-H group?
(b) Based on your answer to part (a), which would you predict to have the higher boiling point, 1-butanol or 1-butanamine?
OH
Amide
H-C-0 H
100
(C)
(a)
TRUE OR FALSE
(a) There are three amines with the molecular formula C3H9N.
(b) Aldehydes, ketones, carboxylic acids, and esters all contain a carbonyl group.
(c) A compound with the molecular formula of C3H6O may be either an aldehyde, a ketone, or a carboxylic acid.
(d) Bond angles about the carbonyl carbon of an aldehyde, a ketone, a carboxylic acid, and an ester are all approximately 109.5°.
(e) The molecular formula of the smallest aldehyde is C3H6O, and that of the smallest ketone is also C3H6O.
(f) The molecular formula of the smallest carboxylic acid is C2H4O2.
Chapter 3 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)
Ch. 3.1 - Prob. 1PCh. 3.2 - (a) Classify the carbon atoms in each compound as...Ch. 3.2 - Problem 3.3 Classify a carbon atom by the number...Ch. 3.2 - Classify each alkyl halide and alcohol as , or...Ch. 3.2 - Prob. 5PCh. 3.2 - Prob. 6PCh. 3.2 - Draw the structure of a compound of molecular...Ch. 3.2 - Prob. 8PCh. 3.2 - Prob. 9PCh. 3.2 - Draw the structure of a compound fitting each...
Ch. 3.4 - Predict which compound in each pair has the higher...Ch. 3.4 - Prob. 17PCh. 3.4 - a Label the hydrophobic and hydrophilic portions...Ch. 3.5 - Prob. 21PCh. 3 - 3.29
Identify the functional groups in the...Ch. 3 - Prob. 32PCh. 3 - 3.31 For each alkane: (a) classify each carbon...Ch. 3 - 3.32 Identify the functional groups in each...Ch. 3 - 3.33 Identify each functional group located in the...Ch. 3 - 3.34 (a)Identify the functional groups in...Ch. 3 - Draw seven constitutional isomers with molecular...Ch. 3 - Prob. 38PCh. 3 - Prob. 39PCh. 3 - Prob. 40PCh. 3 - Intramolecular force of attraction are often...Ch. 3 - 3.40 (a) Draw four compounds with molecular...Ch. 3 - 3.41 Rank the compounds in each group in order of...Ch. 3 - Explain why CH3CH2NHCH3 has higher boiling point...Ch. 3 - Prob. 45PCh. 3 - 3.44 Rank the following compounds in order of...Ch. 3 - Prob. 47PCh. 3 - 3.50 Predict the solubility of each of the...Ch. 3 - Prob. 52PCh. 3 - Prob. 53PCh. 3 - 3.53 THC is the active component in marijuana, and...Ch. 3 - Prob. 55PCh. 3 - Prob. 56PCh. 3 - 3.60 Quinapril (trade name Accupril) is a drug...Ch. 3 - 3.61 Answer each question about oxycodone, a...Ch. 3 - Prob. 65PCh. 3 - Prob. 66PCh. 3 - 3.64 Explain why A is less water soluble than B,...Ch. 3 - 3.65 Recall from section 1.10B that there is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (a) Draw the structures for the eight constitutional isomers of molecular formula C 4H 11N. (b) Give the systematic name for each amine. (c) Identify the chirality center present in one of the amines.arrow_forwardDraw the structure of a compound of molecular formula C 9H 11NO that contains a benzene ring and a: (a) 1 ° amide; (b) 2 ° amide; (c) 3 ° amide.arrow_forwardJj.166. Draw each amide. N-butyl-N-methylbenzamidearrow_forward
- Write the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forward6. Phenobarbital is a long-acting sedative, hypnotic, and anticonvulsant. a) Name all functional groups in this compound. b) Draw structural formulas for the products from complete hydrolysis of all amide groups in aqueous NaOH. H -N N H Phenobarbitalarrow_forwardwhy n-hexanol has higher boiling point than n-hexane. why acetic acid is more acidic then butanoic acid. why sugar is soluble in water in terms of their molecular interactions.arrow_forward
- The following amines have the same molecular formula (C5H13N), but their boiling points are significantly different. Explain why. H `NH2 2-Methylbutan-1-amine Boiling point = 97 °C N-Methylbutan-2-amine Boiling point = 84 °C N-Ethyl-N-methylethan-1-amine Boiling point = 65 °Carrow_forwardDraw the hydrogen bonding that takes place between(a) two molecules of ethanol.(b) two molecules of propylamine.(c) a molecule of dimethyl ether and two molecules of water.(d) two molecules of trimethylamine and a molecule of water.arrow_forwardAmines with more than 6 carbons are soluble in: a) aqueous HCI b) aqueous NaHCO3 d) water c) aqueous NaOH Which of the following would give a positive iodoform test? acetone a) benzophenone c) 3-pentanone d) cyclopentanone meth "Saponification" as the term is used in organic chemistry means: a) acidic hydrolysis of an ester b) basic hydrolysis of an ester c) acidic hydrolysis of an amide d) basic hydrolysis of an amide 3. ( Propylamine can be synthesized by the LiAlH4 reduction of: a) CH3CH2CECH b) CH3CH=NH d)) CH3CH2CEN c) CH3CH2NO2arrow_forward
- A) Name the following amine. H3C−CH2−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound. B ) Name the following amine. CH3−CH2−NH−CH2−CH2−CH3 Spell out the full name of the compound.arrow_forwardОН 1) NaH 2) O 3) Н2Оarrow_forward4) The IUPAC name for the following compound is -- NH2 3-methyl-2-propylpentan-1-amine 2-methyl-3-propylpentan-1-amine O 3-methyl-2-propylhexan-1-amine 2-methyl-3-propylhexan-1-aminearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY