
Concept explainers
(a)
Interpretation:
Current follower that will produce a 1.0 V output for 10.0 µA input current should be designed.
Concept introduction:
Operational amplifiers can be used to measure or process currents by connecting them in the current follower mode. This mode provides a nearly zero resistance load to the current source and prevents it from being loaded by a measuring device or circuit.
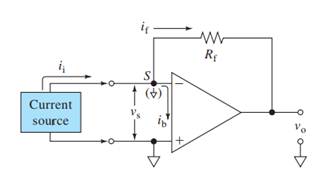
Here,
V+, V- = input voltages
Vs = Input difference voltage
V0 = output voltage
ib = input bias current
if = feedback current
ii = input current
Rf = feedback resistor
Also,
(b)
Interpretation:
Effective input resistance of the current follower designed in part (a) should be calculated.
Concept introduction:
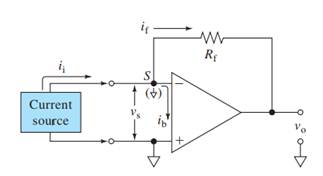
Here,
V+, V- = input voltages
Vs = Input difference voltage
V0 = output voltage
ib = input bias current
if = feedback current
ii = input current
Rf = feedback resistor
Ri = effective input resistance
A = open loop gain
Also,
(c)
Interpretation:
The percent relative error for the circuit designed in part (a) for an input current of 25 µA should be calculated.
Concept introduction:
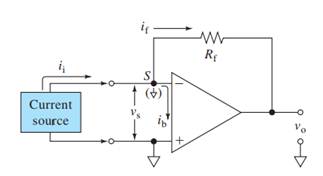
Here,
V+, V- = input voltages
Vs = Input difference voltage
V0 = output voltage
ib = input bias current
if = feedback current
ii = input current
Rf = feedback resistor
Ri = effective input resistance
A = open loop gain
Here,
Also,
Thus,
Relative error percentage =
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Principles of Instrumental Analysis, 6th Edition
- What is the purpose of waiting 1s after a voltage pulse before measuring current in sampled current voltammetry?arrow_forwardElectrochemical detection is commonly used in combination capillary electrophoresis systems. This is a type of hydrodynamic voltammetry called flow injection, where it has been shown that ilim a U1/3, where U is the flow rate of the solution. Flow analysis of a urine sample at a thin-layer amperometric detector, with a flow-rate of 1.25mL/min, yielded a limiting current value of 1.6µA for its unknown uric acid content. A larger current of 2.4µA was observed for a sample containing 1 × 10-4 M uric acid and flowing at a rate of 0.9mL/min. Calculate the original concentration of uric acid in the samplearrow_forwardThe table shows a series of standard additions of Cu(II)to acidified tap water samples measured by anodic stripping voltammetry at a platinum electrode. The unknown and all standard additions were made up to the same final volume. 1. Plot absorbance as a function of the concentration of Cu (II) standard. Determine the equation of the line and the X intercept. Find the concentration of Cu(II) in the water.arrow_forward
- 7) A 25.0 ml sample containing Cu gave an instrument reading of 23.6 units (corrected for a blank). When exactly 0.500 ml of 0.0287M Cu(NO3)2 Was added to the solution,the signal increased to 37.9 units. Calculate the molar concentration of Cu" in the sample +2,arrow_forward(a) A silicon sample maintained at T = 300 K is uniformly doped with Nd = 1016cm-3 donors. Calculate the resistivity of the sample.arrow_forwardA standard solution was prepared containing 10.0 ppm of an analyte and 15.0 ppm of an internal standard. Analysis of the sample gave signals for the analyte and internal standard of 0.155 and 0.233 (arbitrary units), respectively. Sufficient internal standard was added to an unknown sample to make it 15.0 ppm in the internal standard. Analysis of the unknown sample yielded signals for the analyte and internal standard of 0.274 and 0.198, respectively. Calculate the concentration of analyte in the unknown sample.arrow_forward
- A pH probe has a range of 2 to 10 pH units, and a transmitter converts the pH units into a 4 to 20 mA signal. (a) What are the zero and span for the probe? (b) Determine the gain of the probe/transmitter unit. (c) Determine the transmitter output if the pH of a solution is 5.0. (d) If the transmitter gives a reading of 11 mA, what is the pH of the solution?arrow_forwardThe molar absorptivity for aqueous solutions of phenol at 211 nm is 6.17 × 103 L cm-1 mol-1. Calculate the permissible range of phenol concentrations if the transmittance is to be less than 85% and greater than 7% when the measurements are made in 1.00-cm cells.arrow_forwardFIGURE 24-8 2µF 4µF HH 3µF HH 6µF 90V A system of capacitors is connected across a 90 V DC voltage source as shown in Fig. 24-8. What is the equivalent capacitance of this system?arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
