
Concept explainers
(a)
Interpretation:
The
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
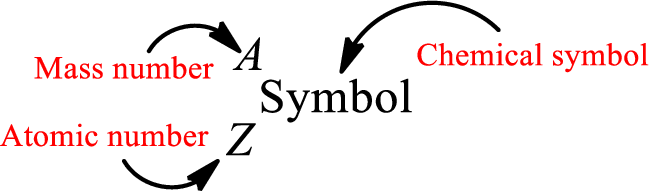
An element is a pure substance that cannot be broken by ordinary
(b)
Interpretation:
The atomic number of atom that has 7 electrons, 8 neutrons, and 7 protons has to be given.
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
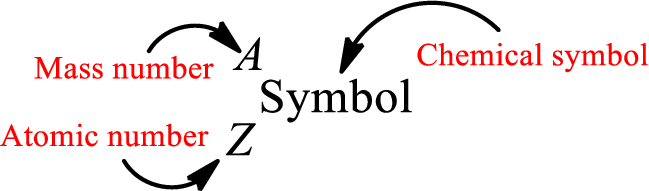
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(c)
Interpretation:
The atomic number of atom that has 13 protons, 14 neutrons and 13 electrons has to be given.
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
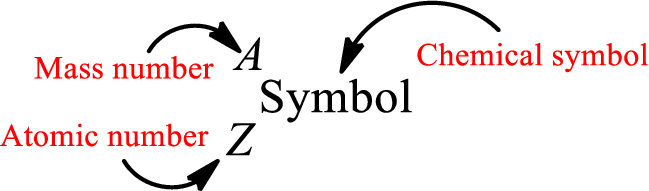
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(d)
Interpretation:
The atomic number of atom that has 28 neutrons, 20 electrons, and 20 protons has to be given.
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
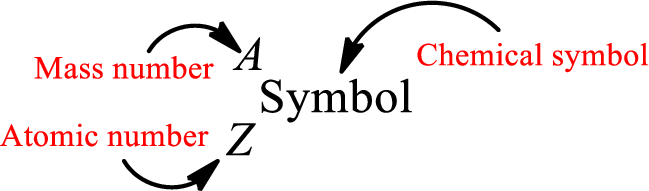
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- An isotope of sodium (Na) has a mass number of 22. How many neutrons does it have? a. 11 b. 12 c. 22 d. 44arrow_forwardWhich element has only one proton?arrow_forwardA sodium atom has 11 protons and 12 neutrons. What is the atomic number of sodium? Multiple Choice A. 34 B. 12 C. 23 D. 11arrow_forward
- 1. 2. 3. 4. 5. 6. 7. What are the top elements that make up 96% of the human body? The image here shows the periodic table designation for Magnesium. a. What is the atomic mass? b. What is the atomic number? c. Draw the atomic structure and label protons, neutrons and electrons. 12 Mg 24.305 Compare and contrast the three types of bonds: Ionic Bond Covalent Bond Hydrogen Bond How do the electrons interact in each type of bond? Define pH and explain how it is measured. What is the difference between a base and an acid? List and EXPLAIN the four important characteristics of water that allow it to sustain life? Compare and contrast dehydration synthesis and hydrolysis. Draw a diagram on the formation and breakdown of the Disaccharide Sucrose to show how this occurs.arrow_forwardWhich of the following levels in the shell model of an atom can hold up to two electrons? A. The second shell B. The first shell C. The third shellarrow_forwardWhich of the following items are pure elements—that is, made up of only one type of atom? Check all that apply. A. Iron B. Cake C. Carbonarrow_forward
- A carbon atom contains six protons and six neutrons.A. What are its atomic number and atomic weight?B. How many electrons does it have?C. How many additional electrons must it add to fill its outermost shell? How does this affect carbon’s chemical behavior?D. Carbon with an atomic weight of 14 is radioactive. How does it differ in structure from nonradioactive carbon? How does this difference affect its chemical behavior?arrow_forwardhow many hydrogen can be formed from the question abovearrow_forwardAtoms are incredibly tiny, so small that most microscopes can't even see them, but atoms themselves are made of even smaller particles: protons, neutrons, and electrons. Which of the following describes where protons are located in an atom? A. A shell B. The nucleus C. Both the nucleus and a shellarrow_forward
- How many protons are present in the atomic nucleus of nitrogen (based on the atomic number)? A. 7 protons 14 protons 21 protons 35 protons 42 protonsarrow_forwardDistinguish atomic number from mass number.arrow_forwardA metal ion with a 2+ charge has 23 electrons and forms a compound with a halogen ion that contains 17 protons. a. What is the identity of the metal ion? b. What is the identity of the halogen ¡on and how many electrons does it contain? c. Determine the compound that it forms and name it.arrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning


