
(a)
Interpretation:
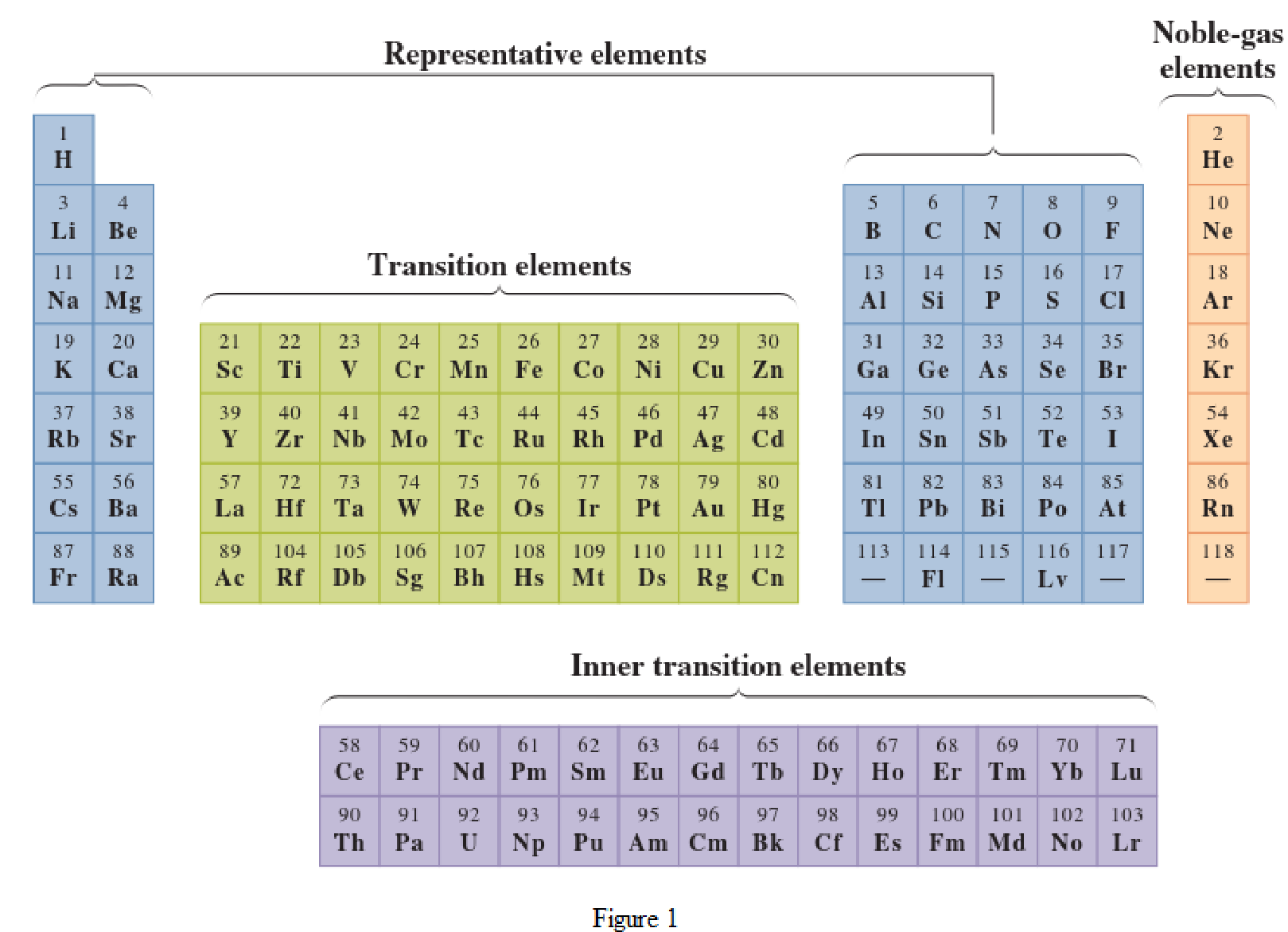
In the given periodic table, how many elements those are highlighted which represent inner
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(b)
Interpretation:
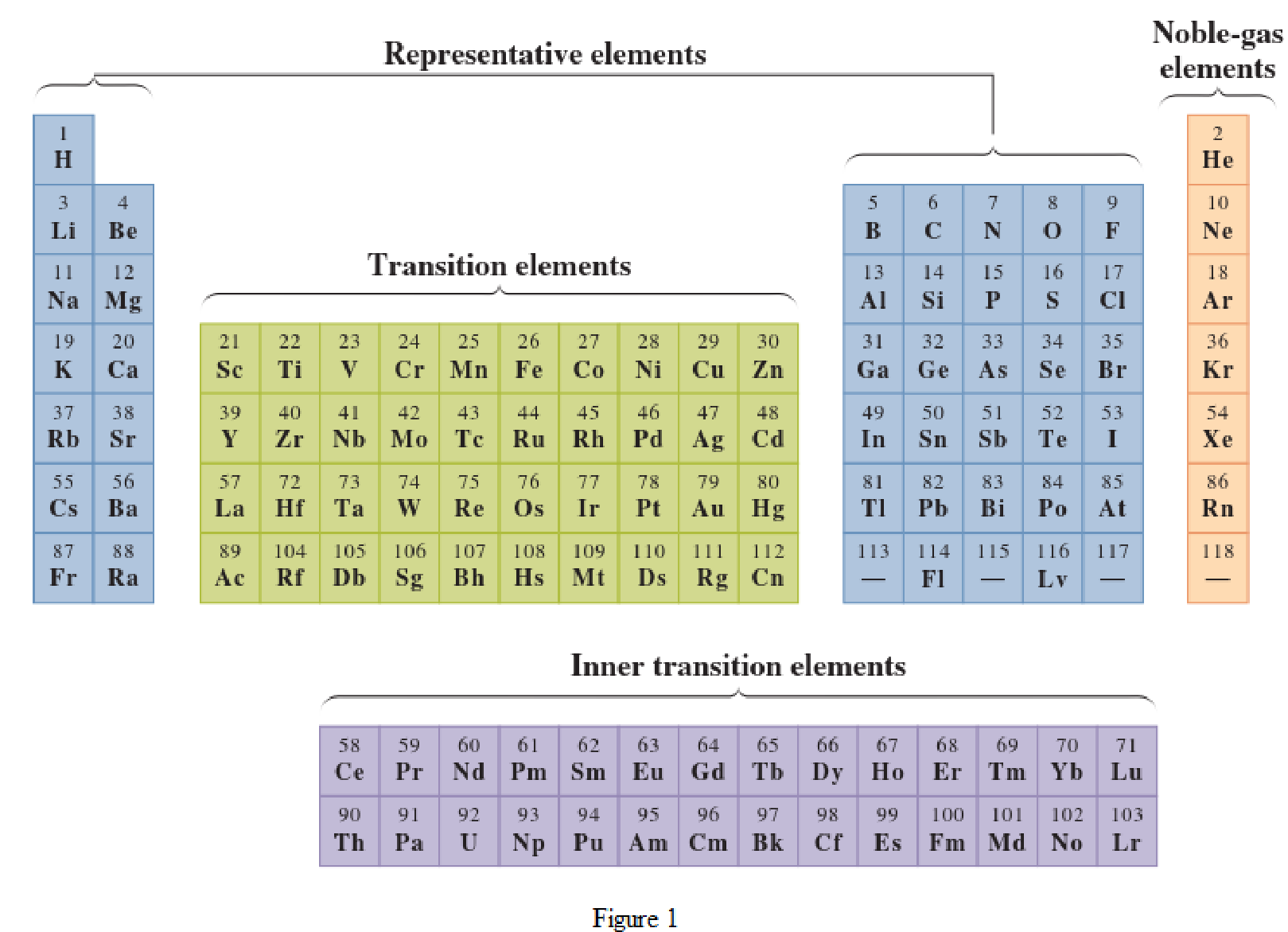
In the given periodic table, how many elements those are highlighted which represent transition have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(c)
Interpretation:
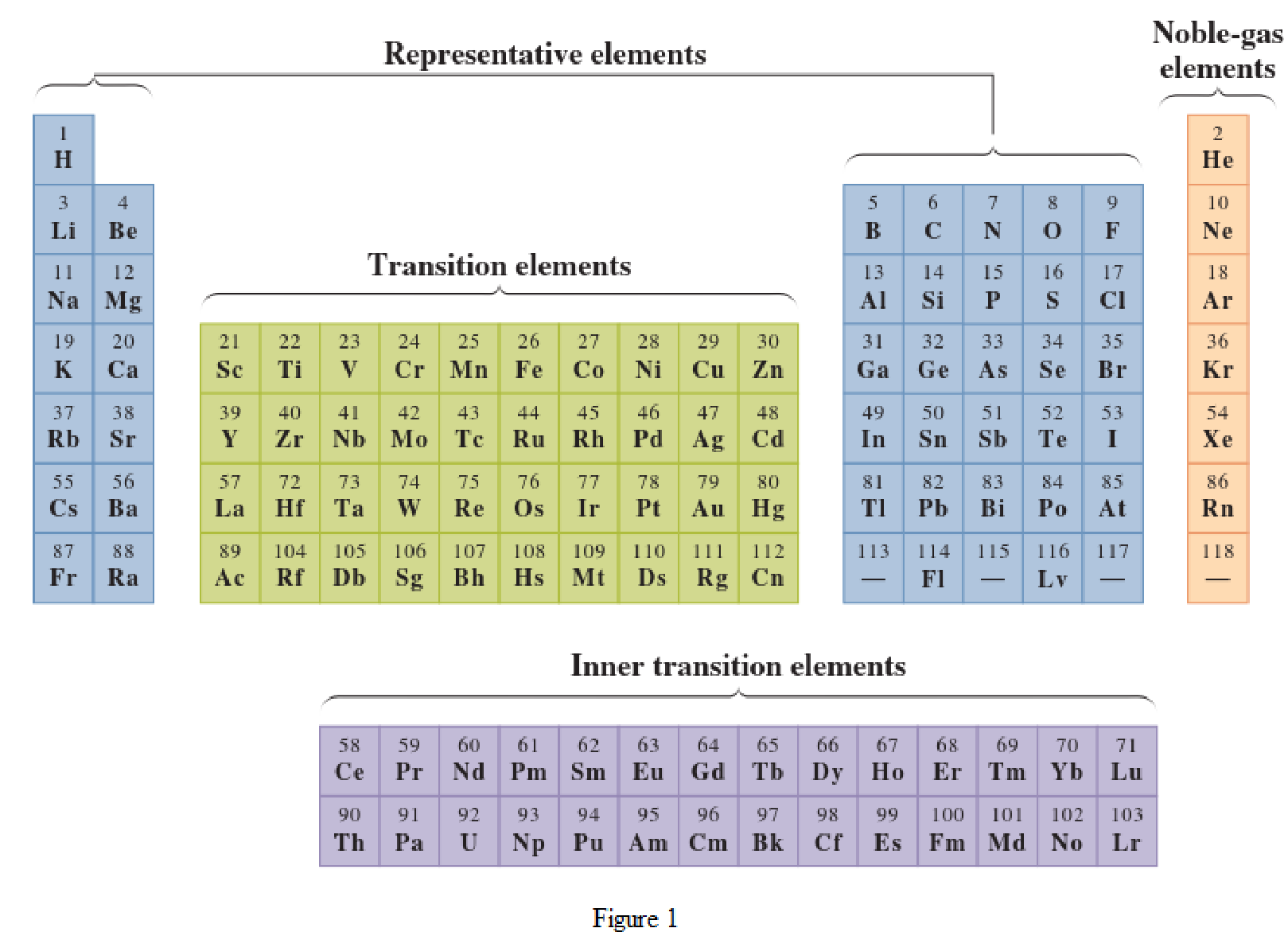
In the given periodic table, how many elements those are highlighted which represent metallic representative elements have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
(d)
Interpretation:
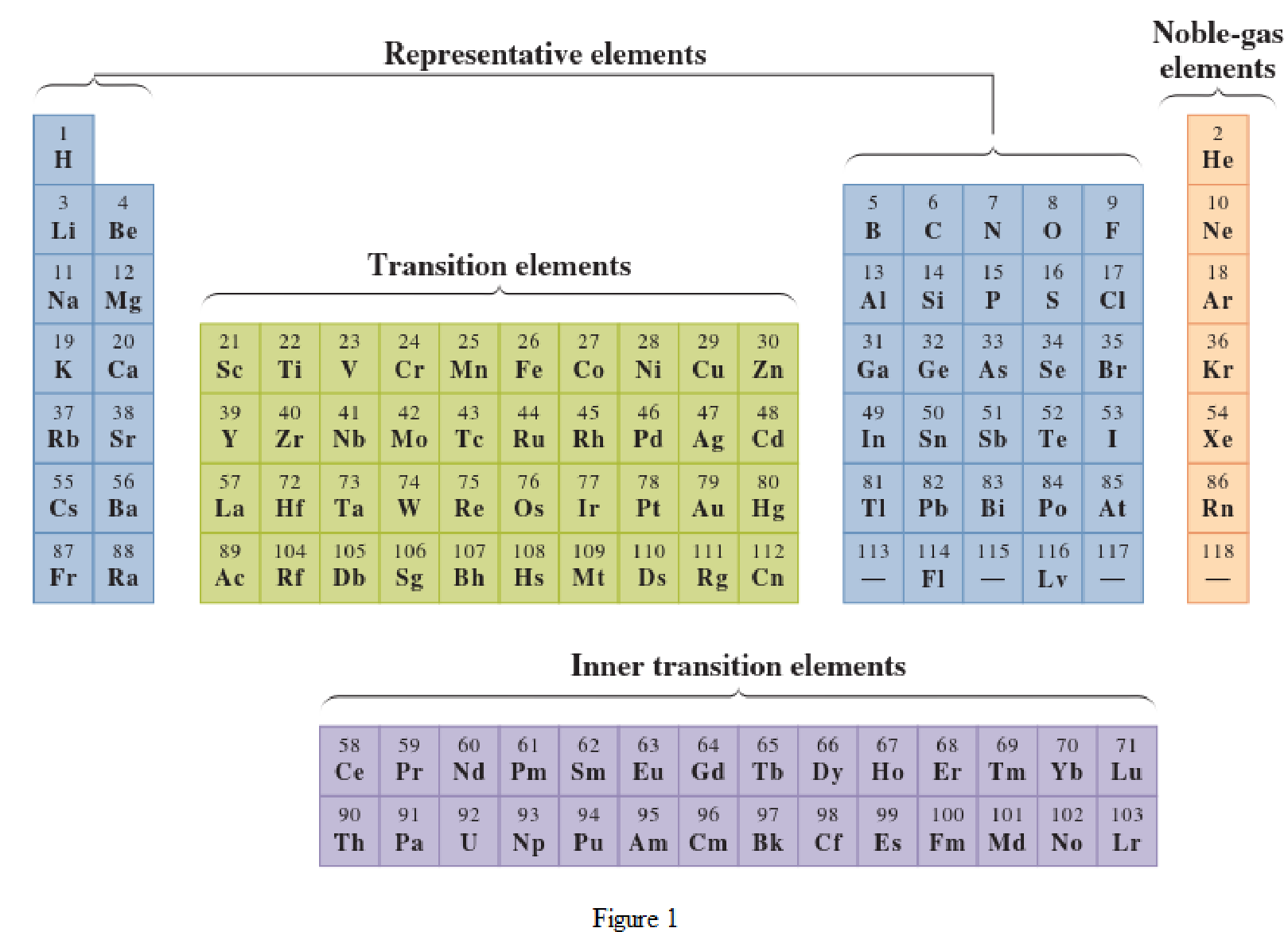
In the given periodic table, how many elements those are highlighted which represent nonmetals have to be determined.
Concept Introduction:
Elements in the periodic table are classified in several different ways and out of them two most common systems are,
- System based on the physical properties in which they are classified as metals and nonmetals.
- System based on electronic configuration in which they are classified as noble-gas, representative elements, transition elements, or inner-transition elements.
Noble-gas elements are the ones that are located in far right of periodic table. The physical state of these elements at room temperature is gas. The noble gases have their electronic configuration ending with
Representative elements are the ones that are in s area and area of the periodic table. They have partially filled s subshell or p subshell in their electronic configurations. Some of the elements are nonmetals while others are metals.
Transition elements are the ones that are located in d area of periodic table. They have the distinguishing electrons in their d subshell. All the transition elements are metals.
Inner transition elements are the ones that are located in f area of the periodic table. They have the distinguishing electrons in their f subshell. All inner transition elements are metals.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Ferrous sulfate is one dietary supplement used to treat iron-deficiency anemia. What are the molecular formula and molecular mass of this compound? howmany milligrams of iron are in 250 mg of ferrous sulfate?arrow_forwardConsider these compounds: A. PbBr, B. MnS C. Ag,CO3 D. AIPO, Complete the following statements by entering the letter(s) corresponding to the correct compound(s). (If more than one compound fits the description, include all the relevant compounds by writing your answer as a string of characters without punctuation, e.g, ABC.) Without doing any calculations it is possible to determine that magnesium fluoride is more soluble than and magnesium fluoride is less soluble than| It is not possible to determine whether magnesium fluoride is more or less soluble than by simply comparing Kgp values.arrow_forwardDetermine the mass in grams of 2.10 × 10²¹ atoms of copper. (The mass of one mole of copper is 63.55 g.)arrow_forward
- A compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of approximately 90 g. What is the molecular formula of the compound?arrow_forwardWork out the following problems: a. What is the pH of a solution with a concentration of0.00001 moles/ml (M) of OH−?b. Draw the atomic structure of magnesium and predict what kindsof bonds it will make.c. What kind of ion would you expect magnesium to make on thebasis of its valence?arrow_forwardThe molecular weight of ethanol (CH3CH2OH) is46 and its density is 0.789 g/cm3.A. What is the molarity of ethanol in beer that is 5%ethanol by volume? [Alcohol content of beer varies fromabout 4% (lite beer) to 8% (stout beer).]B. The legal limit for a driver’s blood alcohol contentvaries, but 80 mg of ethanol per 100 mL of blood (usuallyreferred to as a blood alcohol level of 0.08) is typical. Whatis the molarity of ethanol in a person at this legal limit?C. How many 12-oz (355-mL) bottles of 5% beer coulda 70-kg person drink and remain under the legal limit? A70-kg person contains about 40 liters of water. Ignore themetabolism of ethanol, and assume that the water contentof the person remains constant.arrow_forward
- 75 mL of 0.300 mol/L sodium phosphate solution is combined with 67.5 mL of 0.350 mol/L calcium bicarbonate. a)Before you begin your reaction, you must accurately produce 1.500 L of your sodium phosphate solution from sodium phosphate trihydrate solid. Write out a procedure to explain all the steps you will take in the lab when making the solution to ensure that your solution concentration is accurate. Please include calculations that show the required mass of solid. Also include the correct names of all equipment used. b)You have a super powerful microscope in your lab! You are able to zoom in on your sodium phosphate solution and take a picture at the molecular level. Label the diagram on the left with the correct choices from the box on the right. You may use arrows or rewrite the symbols in one appropriate place. c)In one sentence, explain what the diagram is showing.arrow_forwardDerive the Henderson – Hasselbach Equation. [H*][A¯] Ка— [НА] Molecular Weight: H = 1 g/mol C= 12 g/mol N=14 g/mol O = 16 g/mol Cl= 35.45 g/molarrow_forwardThe liquids butan-1-ol and butanal have similar molar masses. Which is expected to have the higher boiling point? Explain your choices.arrow_forward
- What mass of gallium oxide, Ga2O3, can be prepared from 29.0 g of gallium metal? The equation for the reaction is 4Ga + 3O2 ⟶ 2Ga2 O3.arrow_forwardThe Ksp values of silver chromate Ag2CrO4 and silver iodate Ag(IO3) are given below. Ag2CrO4 Ag(IO3) Ksp 1.12 x 10-12 3.17 x 10-8 Based on these Ksp values, which of the following is true? Choose one option only. Options: a. In the solution consisting of 1.00 x10-4 M Ag+ and 5.00 x10-5 M CrO42-, Ag2CrO4 precipitate will form. b. In the solution consisting of 1.0 x10-4 M Ag+ and 1.0 x10-4 M IO3-, Ag(IO3) precipitate will form. c. In pure water, the solubility of Ag2CrO4 is lower than the solubility of Ag(IO3). d. In the solution consisting of 0.200 M CrO42- and 0.200 M IO3-, Ag2(CrO4) will precipitate first if we add Ag+ ions gradually into the above mixture.arrow_forwardFor Be-10, find the: a.) mase defect (in grams) b.) binding energy in kilojoules per mole. mass proton= 1.00728 amu; mass neutron= 1.00867 amu; mass Be-10 = 10.013534679 amuarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
