
Concept explainers
Interpretation:
The given
Concept introduction:
Atom- is the smallest unit of the matter and it consists of three subatomic particles.
Element- an element is consists only one type of atoms.
Compound- when two or more than two different elements are combined together they form a compound.
Ion- when the electron transfer takes place in an atom, it is termed as ion.
Answer to Problem 61A
In the given reaction the sodium and chlorine atoms are present in their element state, and forming an ionic compound
Explanation of Solution
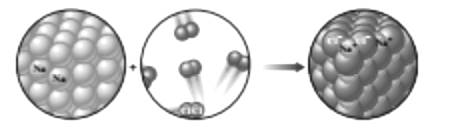
From the given figure following point are comes out-
In any chemical reaction left side substances are reactant and right side substances are product.
The first diagram is showing the sodium element which is consists of sodium atoms and it is solid, where in second chlorine gas is present, the atoms of chlorine are scattered form so it consider as gas.
The sodium and chlorine are combined together and form an ionic compound
The chemical reaction started with the reactant and converted into product. The arrangement or position of atoms explains the element present in solid, liquid or gaseous state.
In the given reaction, the sodium and chlorine atoms are present in their elemental state, and form an ionic compound
Chapter 3 Solutions
World of Chemistry, 3rd edition
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





