
Concept explainers
(a)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
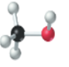
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(b)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
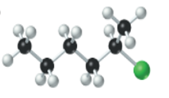
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(c)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
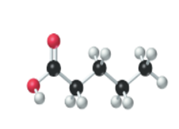
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
(d)
Interpretation:
The name, condensed structural formula and molecular mass of the given molecule should be drawn.
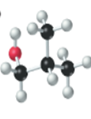
Concept introduction:
Name of the compound can be assigned according to below rule:
First, identify the number of carbon atoms present in chain, and then identify the function group if present in the given compound.
Condensed structural formula is defined as the structural formula which shows the spatial arrangement of bonds in an organic compound. In this structural formula, the bonds between the atoms are shown with lines and omit the bond between carbon and hydrogen atoms.
Molecular mass is defined as the sum of mass of each atom present in the compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Principles and Modern Applications - With Solutions Manual and Modified MasteringChemistry Code
- True or false? The atom with the largest subscript in a formula is die atom with the largest percent by mass in the compound. If true, explain why with an example. If false, explain why with an example. In either case, provide mathematical supportarrow_forwardIn what units are atomic, molecular, and formula mass expressed? Define those units.arrow_forwardIt may be said that because atomic, molecular, and formula masses are all based on carbon-12, they are conceptually alike. What then are their differences?arrow_forward
- True or false? The atom with the largest subscript in a formula is the atom with the largest percent by mass in the compound. If true, explain why with an example. If false, explain why and provide a counter example. In either case, provide mathematical support.arrow_forwardDetermine a chemical formula from elemental analysis (i.e., from % composition).arrow_forwardCalculate the percent by mass of the element listed first in the formulas for each of the following compounds. methane, CH4 sodium nitrate, NaNO3 carbon monoxide, CO nitrogen dioxide, NO2 1-octanol, C8H18O calcium phosphate, Ca3( PO4)2 3-phenyiphenol, C12H10O aluminum acetate, Al(C2H3O2)3arrow_forward
- Questions 27 to 30: Calculate the mass of each substance from the number of moles given. a 0.542mol sodium hydrogen carbonate b 0.0789mol silver nitrate c 9.61mol sodium hydrogen phosphate d 0.903mol calcium bromate e 1.14mol ammonium sulfitearrow_forwardWhich of the three terms atomic mass, molecular mass, or formula mass is most appropriate for each of the following: ammonia, calcium oxide, barium, chlorine, sodium carbonate?arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





