
Study Guide/Solutions Manual for Organic Chemistry
5th Edition
ISBN: 9781259637063
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 31, Problem 31.39P
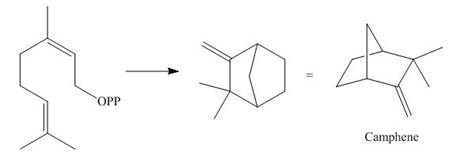
Draw a stepwise mechanism for the following conversion, which forms camphene. Camphene is a component of camphor and citronella oils.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a stepwise mechanism for the following reaction, the last step in a five-step industrial synthesis of vitamin C that begins with the simple carbohydrate glucose.
Draw a stepwise mechanism for the following intramolecular reaction, which is used in the synthesis of the female sex hormone estrone.
Ethers are oxidized with O2 to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.
Chapter 31 Solutions
Study Guide/Solutions Manual for Organic Chemistry
Ch. 31 - Problem 31.1
One component of jojoba oil is a wax...Ch. 31 - Problem 31.2
How would you expect the melting...Ch. 31 - Problem 31.3
Draw the products formed when...Ch. 31 - Problem 31.4
The main fatty acid component of the...Ch. 31 - Prob. 31.5PCh. 31 - Problem 31.6
Draw the structure of a lecithin...Ch. 31 - Prob. 31.7PCh. 31 - Problem 31.8
Why are phospholipids, but not...Ch. 31 - Problem 31.9
Explain why regularly ingesting a...Ch. 31 - Problem 31.10
Locate the isoprene units in each...
Ch. 31 - Problem 31.11
Locate the isoprene units in...Ch. 31 - Problem 31.12
Write a stepwise mechanism for the...Ch. 31 - Prob. 31.13PCh. 31 - Prob. 31.14PCh. 31 - Prob. 31.15PCh. 31 - Prob. 31.16PCh. 31 - 31.17 Locate the isoprene units in each...Ch. 31 - Prob. 31.18PCh. 31 - Prob. 31.19PCh. 31 - Prob. 31.20PCh. 31 - Prob. 31.21PCh. 31 - 31.22 What is the structure of an optically...Ch. 31 - Prob. 31.23PCh. 31 - 31.24 Draw the structure of the following...Ch. 31 - Prob. 31.25PCh. 31 - Locate the isoprene units in each compound. a. e....Ch. 31 - 31.27 Classify each terpene and terpenoid in...Ch. 31 - 31.38 Draw the products formed when cholesterol is...Ch. 31 - 31.29 An isoprene unit can be thought of as having...Ch. 31 - 31.30 Draw a stepwise mechanism for the conversion...Ch. 31 - Prob. 31.31PCh. 31 - Prob. 31.32PCh. 31 - Draw three-dimensional structures f or each...Ch. 31 - Prob. 31.34PCh. 31 - Prob. 31.35PCh. 31 - Prob. 31.36PCh. 31 - Prob. 31.37PCh. 31 - 31.38 Draw the products formed when cholesterol is...Ch. 31 - 31.39 Draw a stepwise mechanism for the following...Ch. 31 - 31.40 Draw a stepwise mechanism for the following...Ch. 31 - Prob. 31.41P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a stepwise mechanism for the attached conversion, which formscamphene. Camphene is a component of camphor and citronella oilsarrow_forwardDraw a stepwise mechanism for the conversion of lactone A to ester Busing HCl in ethanol. B is converted in one step to ethyl chrysanthemate,a useful intermediate in the synthesis of a variety of pyrethrins, naturally occurring insecticides with three-membered rings that are isolated fromchrysanthemumsarrow_forwardDraw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).arrow_forward
- Draw a stepwise mechanism for the nitration of a benzene ring.arrow_forwardA key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.arrow_forwardDraw a stepwise mechanism for the following reaction, one step in the synthesis of the cholesterol-lowering drug ezetimibe.arrow_forward
- Although γ-butyrolactone is a biologically inactive compound, it is converted in the body to 4-hydroxybutanoic acid (GHB), an addictive and intoxicating recreational drug. Draw a stepwise mechanism for this conversion in the presence of acid.arrow_forwardDraw a stepwise mechanism for the conversion of lactone A to ester B using HCl in ethanol. B is converted in one step to ethyl chrysanthemate, a useful intermediate in the synthesis of a variety of pyrethrins, naturally occurring insecticides with three-membered rings that are isolated from chrysanthemums.arrow_forwardTwo naturally occurring compounds that contain stable cyclic hemiacetals and acetals are monensin and digoxin, the chapter-opening molecule. Monensin, a polyether antibiotic produced by Streptomyces cinamonensis, is used as an additive in cattle feed. Digoxin is a widely prescribed cardiac drug used to increase the force of heart contractions. Label each acetal, hemiacetal, and ether in both compounds.arrow_forward
- Draw a stepwise mechanism for the formation of A from an alcohol and acid chloride. A was converted in one step to blattellaquinone, the sex pheromone of the female German cockroach, Blattella germanica.arrow_forwardDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that can be used as a synthetic detergent to clean away dirtarrow_forwardDraw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inammatory drug celecoxib (trade name Celebrex).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY