
Concept explainers
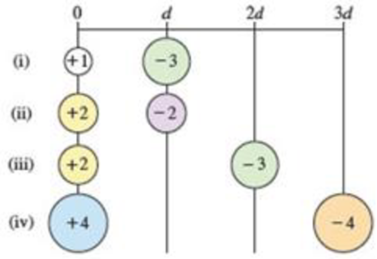
Arrange the following pairs of charged particles in order of increasing magnitude of electrostatic attraction (Eel).
(a) i < ii < iii < iv
(b) iv < iii < ii < i
(c) i = iii < ii < iv
(d) ii < i = iii < iv
(e) iv < i = ii < iii
Interpretation:
The electrostatic potential energies between the given charges in the given statement should be compared by using the concept of electrostatic energy.
Concept Introduction:
Energy is the ability to do work or transfer heat where work is the movement of a body using some force. The SI unit of energy is joule (
Electrostatic energy is potential energy which results from the interaction of charged particles. Oppositely charged particles attract each other and particles of like charges repel each other. The magnitude of the resulting electrostatic potential energy is proportional to the product of the two charges (
If the charges
To calculate: Determine the
Answer to Problem 3.1.3SR
The given pairs of charged particles in order of increasing magnitude of electrostatic attraction (
Explanation of Solution
Reason for correct option
The electrostatic energy between two charges is calculated using the formulae:
It is used to compare the magnitudes of the
For charges +1 and −3:
For charges +2 and −2:
For charges +2 and −3:
For charges +4 and −4:
Therefore, the given pairs of charged particles in order of increasing magnitude of electrostatic attraction (
Reasons for incorrect options:
By doing the calculations in which the given values are substituted, it is found that the option (a) is correct. Therefore, the options (b), (c), (d) and (e) are incorrect.
The electrostatic potential energies between the given charges in the given statement are compared by using the concept of electrostatic energy.
Want to see more full solutions like this?
Chapter 3 Solutions
GEN COMBO CHEMISTRY: ATOMS FIRST; ALEKS 360 2S ACCESS CARD CHEMISTRY:ATOMS FIRST
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
