
Organic Chemistry
10th Edition
ISBN: 9781259253379
Publisher: McGraw Hill Higher Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.15, Problem 19P
Interpretation Introduction
Interpretation:
The most stable conformation of the piperidine derivative is to be drawn. In piperidine derivative the hydrogen bonded to nitrogen has been replaced by methyl.
Concept introduction:
The position of axial and equatorial bonds in cyclohexane chair form is
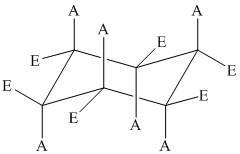
A substituent at an equatorial position is more stable than a substituent at an axial position.
A methyl group has
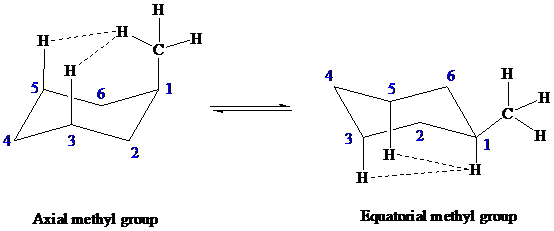
The van der Waals strain is greater between the axial hydrogen and the hydrogen of axial methyl group at
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What type of base should you use?Draw in Newman projection the reactive conformationWhat will be the major product of this reaction? Give the driving mechanism and indicate the stereospecificity using a clear three-dimensional topological structure
Since compounds A and B are in different comformations, how do the products of the elimation reaction differ in conformation between these two compounds? Please provide visual drawing to explain.
Draw the structure of following compound.
(Z)-penta-1,3-diene in the s-trans conformation
Chapter 3 Solutions
Organic Chemistry
Ch. 3.1 - Identify the alkanes corresponding to each of the...Ch. 3.1 - Find the conformations in Figure 3.4 in which the...Ch. 3.2 - Sketch a potential energy diagram for rotation...Ch. 3.2 - Acetylcholine is a neurotransmitter in the central...Ch. 3.2 - Prob. 5PCh. 3.5 - The heats of combustion of ethylcyclopropane and...Ch. 3.8 - Prob. 7PCh. 3.10 - The following questions relate to a cyclohexane...Ch. 3.10 - Draw the most stable conformation of...Ch. 3.11 - Prob. 10P
Ch. 3.11 - Prob. 11PCh. 3.12 - Based on what you know about disubstituted...Ch. 3.12 - Write structural formulas for the most stable...Ch. 3.14 - Cubane (C4H8) is the common name of the polycyclic...Ch. 3.14 - Prob. 15PCh. 3.14 - Prob. 16PCh. 3.14 - Prob. 17PCh. 3.14 - Prob. 18PCh. 3.15 - Prob. 19PCh. 3 - Give the IUPAC names of each of the following: (a)...Ch. 3 - Draw Newman projections for the gauche and...Ch. 3 - Identify all atoms that are (a) anti and (b)...Ch. 3 - Prob. 23PCh. 3 - Prob. 24PCh. 3 - Prob. 25PCh. 3 - Prob. 26PCh. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Oxidation of 4-tert-butylthiane proceeds according...Ch. 3 - The following are representations of two forms of...Ch. 3 - Draw (a) a Newman projection of the most stable...Ch. 3 - Write a structural formula for the most stable...Ch. 3 - Sight down the C-2-C-3 bond, and draw Newman...Ch. 3 - Prob. 34PCh. 3 - Sketch an approximate potential energy diagram for...Ch. 3 - Prob. 36PCh. 3 - Even though the methyl group occupies an...Ch. 3 - Which do you expect to be the more stable...Ch. 3 - Arrange the trimethylcyclohexane isomers shown in...Ch. 3 - Identify the more stable stereoisomer in each of...Ch. 3 - One stereoisomer of 1,1,3,5-tetramethylcyclohexane...Ch. 3 - One of the following two stereoisomers is...Ch. 3 - In each of the following groups of compounds,...Ch. 3 - The heats of combustion of the more and less...Ch. 3 - The measured dipole moment of ClCH2CH2Cl is 1.12D....Ch. 3 - Prob. 46PCh. 3 - Prob. 47PCh. 3 - Prob. 48DSPCh. 3 - Prob. 49DSPCh. 3 - Prob. 50DSPCh. 3 - Prob. 51DSPCh. 3 - Prob. 52DSPCh. 3 - Prob. 53DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw out the complete mechanism (curved arrow formalism) for the preparation of benz-alacetophenearrow_forwardClassify each of the following cycloadditions and explain why orbital symmetry conservation rules allow or forbid each of the following reactions to occur as a concerted process.arrow_forwardCould you show the structural formulas of the compounds A-C, whitch of these compounds is the most likely to form if the reaction occures via a chair conformation intermediate and why?.arrow_forward
- Draw the structure of following compound. (2E,4E)-3-methylhexa-2,4-diene in the s-cis conformationarrow_forwardWhat will be the optical conformation of the product in the following reaction?arrow_forwardDraw the structures of A - D from the following electrocyclic and cycloaddition reactions.arrow_forward
- For each of the following molecules draw the most stable conformations indicated in the parenthesis, and along the specified bonds.a) sec-butylcyclohexane (Newman projection chair along C1-C2 and C5-C4)arrow_forwardFor the following reactions:a. Provide the missing major product, OR, missing reagent and conditions appropriate.b. Pay attention to stereochemistry unless directed otherwise and identify any racemicmixture using the symbol (±)c. Assign each reaction as addition, elimination, substitution or rearrangement, unlessdirected otherwise.arrow_forwardA. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer. B. Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed. -Isomaltose is a reducing sugar while trehalose is not.-Trehalose is very resistant to acid hydrolysis while isomaltose can be acid-hydrolyzed withease.arrow_forward
- Draw the major organic product of each reaction. Indicate the stereochemistry at the stereogenic center. Omit byproducts such as salts. If applicable, expand octets to minimize formal charges.arrow_forwardThe first alkene which requires the position of the double bond to be indicated is?arrow_forwardIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1- phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Whichproduct predominates—the product of inversion or the product of retention of conguration? (c) Suggest an explanation for this phenomenon.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License