
ORGANIC CHEMISTRY W/CONNECT & ALEKS
6th Edition
ISBN: 9781264683888
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.2, Problem 2P
(a) Classify the carbon atoms in each compound as
[1] 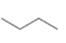 [2]
[2] [3]
[3]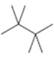 [4]
[4]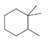
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider the tricyclic structure A. (a) Label each substituent on the rings as axial or equatorial. (b) Draw a skeletal structure for A, using wedges and dashed wedges to show whether the substituents are located above or below the rings.
Consider the tricyclic structure A. (a) Label each substituent on the rings as axial or equatorial. (b) Draw a skeletal structure
for A, using wedges and dashes to show whether the substituents are located above or below the rings.
15) Give the IUPAC name for each compound.
Chapter 3 Solutions
ORGANIC CHEMISTRY W/CONNECT & ALEKS
Ch. 3.1 - Prob. 1PCh. 3.2 - (a) Classify the carbon atoms in each compound as...Ch. 3.2 - Problem 3.3 Classify a carbon atom by the number...Ch. 3.2 - Classify each alkyl halide and alcohol as , or...Ch. 3.2 - Prob. 5PCh. 3.2 - Prob. 6PCh. 3.2 - Draw the structure of a compound of molecular...Ch. 3.2 - Prob. 8PCh. 3.2 - Prob. 9PCh. 3.2 - Draw the structure of a compound fitting each...
Ch. 3.4 - Predict which compound in each pair has the higher...Ch. 3.4 - Prob. 17PCh. 3.4 - a Label the hydrophobic and hydrophilic portions...Ch. 3.5 - Prob. 21PCh. 3 - 3.29
Identify the functional groups in the...Ch. 3 - Prob. 32PCh. 3 - 3.31 For each alkane: (a) classify each carbon...Ch. 3 - 3.32 Identify the functional groups in each...Ch. 3 - 3.33 Identify each functional group located in the...Ch. 3 - 3.34 (a)Identify the functional groups in...Ch. 3 - Draw seven constitutional isomers with molecular...Ch. 3 - Prob. 38PCh. 3 - Prob. 39PCh. 3 - Prob. 40PCh. 3 - Intramolecular force of attraction are often...Ch. 3 - 3.40 (a) Draw four compounds with molecular...Ch. 3 - 3.41 Rank the compounds in each group in order of...Ch. 3 - Explain why CH3CH2NHCH3 has higher boiling point...Ch. 3 - Prob. 45PCh. 3 - 3.44 Rank the following compounds in order of...Ch. 3 - Prob. 47PCh. 3 - 3.50 Predict the solubility of each of the...Ch. 3 - Prob. 52PCh. 3 - Prob. 53PCh. 3 - 3.53 THC is the active component in marijuana, and...Ch. 3 - Prob. 55PCh. 3 - Prob. 56PCh. 3 - 3.60 Quinapril (trade name Accupril) is a drug...Ch. 3 - 3.61 Answer each question about oxycodone, a...Ch. 3 - Prob. 65PCh. 3 - Prob. 66PCh. 3 - 3.64 Explain why A is less water soluble than B,...Ch. 3 - 3.65 Recall from section 1.10B that there is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Dexamethasone is a halogen-containing steroid used to treat infl ammation in rheumatoid arthritis and other conditions. (a) Classify the alkyl halide in dexamethasone as 1 °, 2 °, or 3 °. (b) Classify the hydroxyl groups as 1 °, 2 °, or 3 °.arrow_forward3. Consider the following structures: Br H3C- CH 3 C₂H5 -H -OH Structure A Br HO- H CH3 -CH3 -C₂H5 Structure B C₂H5 The relationship between structure A and B is: The relationship between structure A and C is: The relationship between structure A and D is: The relationship between structure B and C is: Br The relationship between structure B and D is: The relationship between structure C and D is: OH H CH3 CH3 (a) Designate each chirality center as (R) or (S) in structures A, B, C and D. (b) Fill in the blanks for the structural relationship between the compounds in each of the pairs that follow. They may be unrelated, identical (the same compound), constitutional isomers, diastereoisomers, enantiomers. Structure C H₂C H Br 4|||| C₂H5 ||||||CH 3 OH Structure Darrow_forwardexplain why methanethiol, CH3SH, has a lower boiling point (6°C) than methanol, CH3OH (65°C), even though methanethiol has a higher molecular weightarrow_forward
- Question 1 Write the IUPAC name for each compound: сно CHO (a) (b) (e) Question 2 Write the IUPAC name for each compound: H,C CH, (a) CH,-CH(CH,)ẠCH, (b)arrow_forward1) identify the reactant/s in the chemical equation and circle it, also name its major functional group 2) identify the product/s in the chemical equation (circle it) and name its functional group 3) is the a reversible reaction or not? How do we knowarrow_forward(ii) Name the following using IUPAC system of nomenclature: (a) CH2 (Cl) CH (NH2) CHO (b) CH3 C (Br) CH2 CH (F) COOH Brarrow_forward
- OH H3C Type of Reaction: CH3 CH3 CH3 + OH Type of Reaction: + [0] НОarrow_forwardClassify each pair of compounds as constitutional isomers or identical molecules.arrow_forwardClassify the following reaction as: addition, elimination, substitution or rearrangement. A) substitution CH3 B) addition CH3 OTS C) elimination D) rearrangementarrow_forward
- Name each compound. CH; CH, CH, · CH3-CH-CH-CH-CEC-H CH3arrow_forwardAssign (R) or (S) designations to each of the following compounds: (a) (b) HO (c) CH3 .CI H.arrow_forwardCompound A Compound F HCI CH3OH H₂SO4 Compound B CH3 CH₂ CH₂ Cl Compound E NaOH K₂Cr₂O7 H₂SO4 Compound C K₂Cr₂O7 H₂SO4 Compound D a. Draw the structural formulas of compounds A, C, D, E and F in the boxes provided above.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY