
Interpretation:
The total number of neutrons present in the atom that has
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
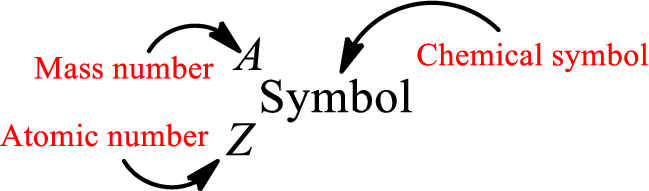
An element is a pure substance that cannot be broken by ordinary
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- How many electrons are present in an atom of P1531? a. 15 b. 16 c. 31 d. no correct responsearrow_forwardExercise 4.3 Give the number of protons, neutrons, and electrons in the atom symbolized by80201Hgarrow_forwardNaturally occurring bromine is a mixture of 50.69% Br-79 with an amu = 78.92 and 49.31% Br-81 with a mass of 80.92 amu. What is the atomic weight of bromine? Show work. (5 points)arrow_forward
- Problem 2-2 What is the mass number of an atom containing: (a) 15 protons, 15 electrons, and 16 neutrons? (b) 86 protons, 86 electrons, and 136 neutrons?arrow_forwardI need answer for Q.N- 23arrow_forwardWhich of these are isotopes of hydrogen?A) 12C and 13C B) 42K and 40K C) Li+ and He D) 3H and 2H E) 2H and HeIs this correct answer D?arrow_forward
- How many unpaired electrons are presents in the orbital diagram for an atom whose electron configuration is1s22s22p4? a. none b. two c. four d. no correct responsearrow_forwardExercise 4.5 Give the symbol and atomic number for each of the following elements. Also indicate whether each element is a metal or a nonmetal and whether it is a member of a named family. a. argon b. chlorine c. barium d. cesiumarrow_forwardWhat is the Minimum Ni-S distance in alpha-NiS to 4 decimal places in Angstroms? What is the Minimum S-S distance in NiS2 to 4 decimal places in Angstroms? please show how to get answer.arrow_forward
- What is the velocity of electrons emitted from K metal (threshold energy = 2.25eV) using incident radiation of 6.2eV. Input only answer for velocity in m/sarrow_forwardQUESTION 3 What is the the frequency (in kHz) of gamma ray radiation that has a wavelength of 0.01516 nanometers? The speed of light is 3.00x10 8 m/s. Key Concept: Frequency, ν, and wavelength, λ, of light are inversely related to each other by c = λ*ν where c is the speed of light.arrow_forwardA Geiger counter operates on the principle that radiation: answer choices A) darkens a photographic film B) can be detected by its odor C) ionizes atoms and molecules D) no correct responsearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning




