
Concept explainers
Interpretation: The given table and figure needs to be completed.
Concept Introduction: Every matter is made of chemicals; the importance of chemistry is that it's the study of everything. Chemistry is all about how something interacts to affect the structure, composition, as well as knowing properties of substances.
Answer to Problem 6RQ
A complete figure is:
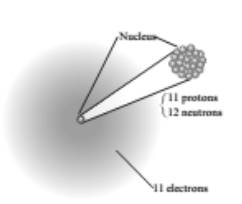
A complete table:
|
| |
| Name of the element | Sodium |
| Symbol of the element | Na |
| Mass number of the atom |
|
| Number of electrons in the atom |
|
Explanation of Solution
Protons are defined as a positively charged
An atom has a nucleus with a positive charge due to its protons and has electrons in the space surrounding the nucleus at relatively large distance from it. The structure shown in figure is of sodium. So, complete figure is:
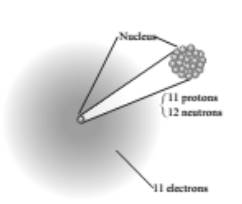
According to this, a complete table:
| Atomic number of an atom |
|
| Name of the element | Sodium |
| Symbol of the element | Na |
| Mass number of the atom |
|
| Number of electrons in the atom |
|
Chemical representation of Sodium is Na. Atomic number of an
Chapter 3 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





