
Concept explainers
Interpretation:
The number of different orbitals that contain electrons for the atom that has electronic configuration as
Concept Introduction:
Electronic configuration of an atom describes how many electrons are present in the shell. Many orbitals are present about the nucleus of an atom. In these orbitals the electrons do not occupy randomly. There are three rules for assigning the electrons to various shells, subshells, and orbitals. They are,
- The subshells are filled in increasing order of energy.
- In a subshell, the electrons occupy the orbital singly first in all orbitals before pairing up by the second electron. All the electrons that are in singly occupied orbitals have same spin.
- In a given orbital there cannot be more than two electrons and they have opposite spins.
Electronic configuration of an element is the one that gives information about how many electrons are present in each electron subshell of an atom. The electrons are added to the subshells in increasing order of energy. Electronic configurations are written in shorthand notation which uses a number‑letter combination. The shell is indicated by the number and subshell is indicated by the letter. Superscript that follows the subshell tells how many electrons are present in the subshell.
The order of filling up the electrons in the subshell is done as shown in the given figure below.
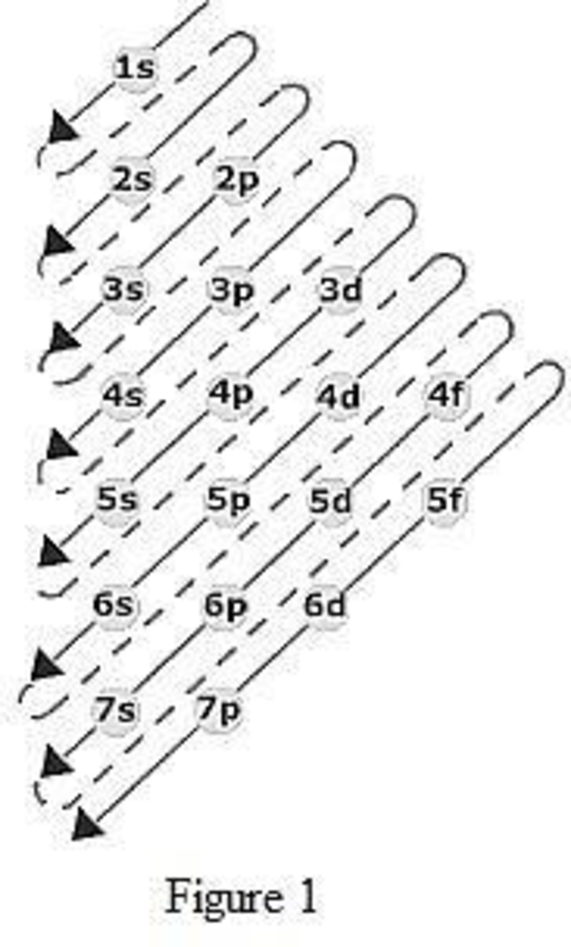
Orbital diagram is the one that gives information about the number of electrons present in the electron orbitals of an atom. The difference between electronic configuration and orbital diagram is that electronic configuration deals with the electrons occupancy in the subshell while orbital diagram deals with the electrons occupancy in the electron orbital. Electrons are paired up in the orbital only when all the orbitals in the subshell are singly filled.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- Which of the following statements is consistent with the electron configuration 1s22s22p3? a. There are 3 electrons in the 2p orbital. b. There are 3 electrons in the 2p subshell. c. There are 3 electrons in the 2p shell. d. no correct responsearrow_forwardHow many electrons can a 3d subshell accommodate? a. 2 b. 6 c. 10 d. no correct responsearrow_forwardThe maximum number of electrons that an electron orbital can accommodate a. is two b. is six c. varies, depending on the type of orbital d. no correct responsearrow_forward
- An electron configuration is a statement of how many electrons an atom has in each of its a. electron shells b. electron subshells c. electron orbitals d. no correct responsearrow_forwardWhich of the following combinations of n, l, ml and ms are correct? Can be more than 1 answer, 1. n = 2, l = 2, ml = 0, ms = -1/2 2. n = 2, l = 1, ml = 0, ms = 3/2 3. n = 4, l = 3, ml = 2, ms = 1/2 4. n = 2, l = 0, ml = 0, ms = -1/2arrow_forwardThe nucleus of an atom contains:A. electrons and protons only.B. protons only.C. electrons, protons, and neutrons.D. protons and neutrons only.Reset SelectionMark for Review What’s This?arrow_forward
- 1) Write the full and noble gas configuration for Se. Full Electron Configuration: Response Noble Gas Configuration: 2) The element represented by the following electron configuration is ____________. [Rn] 7s2 5f11 A)Rb B) Es C) Au D)Cf 3)Write the full and noble gas configuration for Cs. Full Electron Configuration: Noble Gas Configuration: please answer the three question correct pleasearrow_forwardA local AM radio station broadcasts at an energy of 4.20×10-31 kJ/photon.Calculate the frequency at which it is broadcasting.Frequency = KHz(1 KHz = 103 sec -1) Submit Answerarrow_forwardHow many electrons are present in an atom of P1531? a. 15 b. 16 c. 31 d. no correct responsearrow_forward
- Problem 10: Consider the de Broglie wavelength of an electron. What is the de Broglie wavelength of an electron travelling at a speed of 5.0×106 m/s? Give your answer in pm.arrow_forwardIve tried 3d^7 also i tried 3d^1,3d^2,3d^3,3d^4,3d^5,3d^7 and all come back wrongarrow_forwardWrite an orbital diagram for the ground state of the phosphorus atom (Z = 15). Write all orbitals.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

