
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305864504
Author: Brent L. Iverson, Sheila Iverson
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.8, Problem CQ
The amino acids cysteine and serine are shown. What are the Cahn-Ingold-Prelog stereochemical descriptors for these two amino acids?
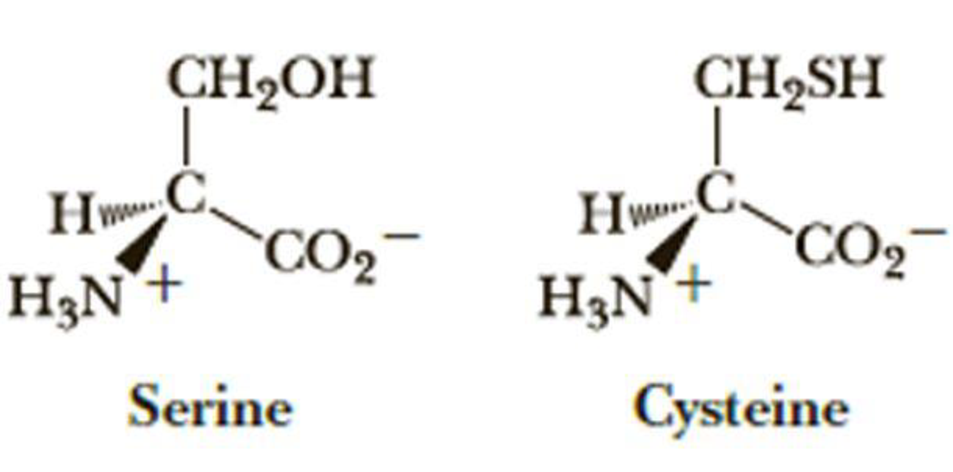
- 1. Serine is S while cysteine is R.
- 2. Cysteine is S while serine is R.
- 3. Cysteine and serine are both R.
- 4. Cysteine and serine are both S.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Dokmarks Profiles Tab Window Help
World view x Paraphras x
n/takeAssignment/takeCovalentActivity.do?locator-assignment-take
X
Bastery
s 2req
D
ats 2req
pts 2req
pts 2req
pts 2req
1 pts 2req
1 pts 2req
1 pts 2req
1 pts 2req
Homework X
-
1 pts 2req
. 1 pts 2req
2
World view.pdf
#
80
30 F3
What is the charge on the ion?:
What is the name of the element?
An ion from a given element has 37 protons and 36 electrons.
What is the symbol for the ion?
$
1
Submit Answer
[Review Topics]
[References]
Use the References to access important values if needed for this question.
Q
F4
SEP
14
%
5
Retry Entire Group
9
Grammar X
tv
F5
<6
Ⓡ
♫
9 more group attempts remaining
MacBook Air
C F6
&
7
My Home X
2.1
A
all/AO
F7
*
8
∞
F
FB
OWLv2 10 X
61
9
F9
*
10
+
@
9
Show All
F10
Q
⠀
X
9
W
.
4)
Wed Sep 14 1:17 PM
F11
+ 11
F12
. Identify the following antioxidant molecule. A. alpha tocopherol hydroquinone
dehydroascorbic acid
CH3 CH3 C. alpha tocopherol semi-quinone
\ / D. ascorbic acid
C = C E. alpha tocopherol quinone
/ \
: O – C C – O CH3
\\ // \ /
C – C C – CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH2–CH2–CH2–CH–CH3
/ \ / l l l
CH3 CH2– CH2 CH3 CH3 CH3
Which of the following will exhibit cis-trans isomerism?
Fe°: [Ar]4s?3d6
Cdº: [Kr]5s²4d10
Pt0: [Xe]ós²5d®
O A. [FeBr2Cl2]2
O B. [Cd(NH3)2(H20)2]2+
O C. [PtBr2(NH3)2]
O D. All of the above.
Chapter 3 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 3.2 - Prob. 3.1PCh. 3.3 - Assign priorities to the groups in each set. (a)...Ch. 3.3 - Prob. 3.3PCh. 3.4 - Following are stereorepresentations for the four...Ch. 3.4 - Prob. 3.5PCh. 3.4 - Prob. 3.6PCh. 3.5 - How many stereoisomers exist for...Ch. 3.5 - How many stereoisomers exist for...Ch. 3.7 - Prob. 3.9PCh. 3.7 - Prob. 3.10P
Ch. 3.8 - If the side chain of the amino add is a methyl...Ch. 3.8 - Prob. BQCh. 3.8 - The amino acids cysteine and serine are shown....Ch. 3.8 - Prob. DQCh. 3.8 - As stated, proteins are stereochemically pure...Ch. 3.8 - As stated, proteins are stereochemically pure...Ch. 3 - Prob. 3.11PCh. 3 - One reason we can be sure that sp3-hybridized...Ch. 3 - Which compounds contain chiral centers? (a)...Ch. 3 - Prob. 3.15PCh. 3 - Prob. 3.16PCh. 3 - Prob. 3.17PCh. 3 - Mark each chiral center in the following molecules...Ch. 3 - Prob. 3.19PCh. 3 - Assign priorities to the groups in each set. (a) H...Ch. 3 - Following are structural formulas for the...Ch. 3 - Following is a staggered conformation for one of...Ch. 3 - Prob. 3.23PCh. 3 - When oxaloacetic acid and acetyl-coenzyme A...Ch. 3 - Prob. 3.25PCh. 3 - Mark each chiral center in the following molecules...Ch. 3 - Prob. 3.27PCh. 3 - Prob. 3.28PCh. 3 - Prob. 3.29PCh. 3 - Prob. 3.30PCh. 3 - Which of the following are meso compounds?Ch. 3 - Prob. 3.32PCh. 3 - Prob. 3.33PCh. 3 - Which of the following compounds are chiral?...Ch. 3 - Prob. 3.35PCh. 3 - Prob. 3.36PCh. 3 - Prob. 3.37PCh. 3 - The chiral catalyst (R)-BINAP-Ru is used to...Ch. 3 - Prob. 3.39P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Drag and drop the correct terms in front of their definitions (not all terms are used). 1. the ability of a protein to bind one molecule but not another. specificity 2. the part of a protein molecule that binds the ligand. binding site 3. the ability of a protein to alter its shape to it more closely with that of the ligand. saturation induced fit irreversible inhibitionarrow_forwardwhat type of isomers are the compounds? which compound have a pair of enantiomers? pls explain and show the enantiomersarrow_forward3. Draw the structure of the cross-link expected between a tyrosine side chain and the sulfonated diazonium ion shown below, CD C3 ANS H20arrow_forward
- A 1.00-mg sample of a pure protein yielded on hydrolysis 0.0165 mg of leucine and 0.0248 mg of isoleucine. What is the minimum possible molar mass of the protein? (MMleucine=MMisoleucine=131g/mol)arrow_forwardFor alanine, Ka1=5.1105,Ka2=1.81010 . Calculate the ratios [Z]/[C+] and [Z]/[A+] at pH (a) 2.00. (b) 6.00. (c) 10.50. What is the principal species at each pH?arrow_forwarda. Explain why the presence of certain proteins in body fluids such as urine or blood can indicate that cellular damage has occurred in the body. b. Explain why a protein is least soluble in an aqueous medium that has a pH equal to the isoelectric point of the protein.arrow_forward
- Given the structures of cysteine, serine and glycine: H,N-CH- -OH H,N-CH- -OH H,N-CH- -OH ČH2 CH2 SH cysteine serine glycine The pair of amino acids that can form a disulfide bridge in a protein is: a. cysteine and serine. b. cysteine and glycine. c. cysteine and cysteine. O d. serine and serine.arrow_forwardPlease give answer all of sub parts....arrow_forward3. Name the following heterocydic compounda? C. Hz ELarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lanthanoids and its Position in Periodic Table - D and F Block Elements - Chemistry Class 12; Author: Ekeeda;https://www.youtube.com/watch?v=ZM04kRxm6tY;License: Standard YouTube License, CC-BY