
Concept explainers
(a)
Interpretation:
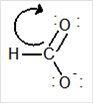
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
(b)
Interpretation:
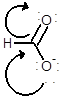
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
(c)
Interpretation:
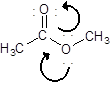
Draw the contributing structure of.
Concept Introduction:
Lewis electron dot symbol is a structure of a molecule that shows the bonding between atoms as well as lone pairs of electrons of atoms also.
Valence electron: The electrons which are present in the outer most energy level are known as valence electron. This can be calculated by the group number of the element. Generally the group number is same as the valence electrons of any elements.
Trending nowThis is a popular solution!

Chapter 3 Solutions
Introduction to General, Organic and Biochemistry
- Problem (#2.) For each ion below, draw all reasonable resonance structures (linked by resonance arrows “↔”). Include curved arrows that indicate the movement of electrons between each resonance structure. Assign non-zero formal charges to each atom for each resonance structure. (a.) NO3– (nitrate) (b.) CH3COO– (acetate) (c.) N3– (azide) (d.) NCO– (isocyanate) Problem (#3.) For each ion in question 2, draw a resonance hybrid, assigning non-zero formal and/or partial charges (δ+, δ–) as needed. Problem (#4.) For each skeletal structure below, satisfy the valences (or octets) of all of the atoms by filling in double and triple bonds as well as unshared electron pairs. Assign non-zero formal charges and show the overall charge if the structure is an ion. See photo attached for Problem number 4. Problem (#5.) For each structure in question 4, draw a resonance hybrid (if it has one) and assign non-zero formal and/or partial charges as needed.arrow_forwardConvert the following representation of ethane, C2H6, into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = C, ivory = H).arrow_forwardWhich of the following are molecular formulas? Select all that apply. Group of answer choices C3H6O3 C2H6O C2H4O2 C3H9O2 CH2Oarrow_forward
- 3-37 Answer true or false. (a) An ionic bond is formed by the combination of positive and negative ions. (b) An ionic bond between two atoms forms by the transfer of one or more valence electrons from the atom of higher electronegativity to the atom of lower electronegativity. (c) As a rough guideline, we say that an ionic bond will form if the difference in electronegativity between two atoms is approximately 1.9 or greater. (d) In forming NaCl from sodium and chlorine atoms, one electron is transferred from the valence shell of sodium to the valence shell of chlorine. (e) The formula of sodium sulfide is Na2S. (f) The formula of calcium hydroxide is CaOH. (g) The formula of aluminum sulfide is MS. (h) The formula of iron(III) oxide is Fe2O2. (j) Barium ion is Ba2, and oxide ion is O2-; there fore, the formula of barium oxide is Ba2O2.arrow_forwardAnswer the following question about esmolol (attached) , a drug used to treat high blood pressure sold under the trade name Brevibloc. Question: Label all C's that bear a δ+ charge.arrow_forward(please show reaekson and incorrect option explain and correct answer) In the major resonance structure for CN^-, what is the formal charge on the N atom? A) 0 B) +2 Q-1 D) +1arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



