
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234722
Author: Klein
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.9, Problem 3.44P
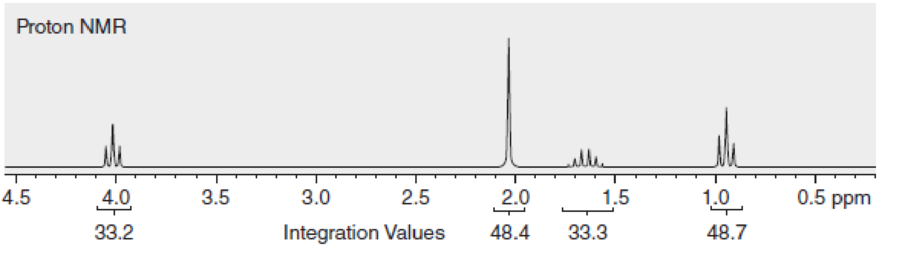
Propose a structure for a compound with molecular formula C5H10O2 that is consistent with the following proton NMR spectrum.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A compound with molecular formula C5H10O2has the following NMR spectrum.
Determine the number of protons giving rise to the signals at 2.0 ppm and 4.0 ppm.
The following NMR spectra were obtained from a compound with the molecular formula C4H6O. Use this information to predict its structure.
Identify the compound with molecular formula C4H9BrO that gives the following 1H NMR spectrum.
Chapter 3 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 3.1 - Prob. 3.2PCh. 3.1 - Prob. 3.3PCh. 3.1 - Prob. 3.4PCh. 3.1 - Prob. 3.5PCh. 3.1 - Prob. 3.6PCh. 3.1 - Prob. 3.7PCh. 3.1 - Prob. 3.8PCh. 3.1 - Prob. 3.9PCh. 3.1 - Prob. 3.10PCh. 3.1 - If you look at your answers to the previous...
Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Prob. 3.19PCh. 3.3 - Prob. 3.21PCh. 3.3 - Prob. 3.22PCh. 3.3 - Prob. 3.23PCh. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.9 - Prob. 3.43PCh. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Prob. 3.48PCh. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
PRACTICE PROBLEM 9.13 How many signals would you expect to obtain in the 1HNMR spectrum of undeca-deuteriocyclo...
Organic Chemistry
The gas-phase reaction between methanol and acetic acid to form methyl acetate and water CH3OH + CH3COOH CH3COO...
Elementary Principles of Chemical Processes, Binder Ready Version
What is the reducing agent in the following reaction?
2 Br –– (aq) + H2 O2 (aq) + 2 H+ (aq) → Br2 (aq) + 2 H2 ...
Chemistry: The Central Science (13th Edition)
Chlorine has two isotopes, 35Cl and 37Cl; 75.77% of chlorine is 35Cl, and 24.23% is 37Cl. The atomic mass of 35...
Organic Chemistry
When a hydrochloric acid solution is combined with a potassium hydroxide solution, an acid-base reaction occurs...
Introductory Chemistry (6th Edition)
50. For each solution, calculate the initial and final pH after adding 0.010 mol of NaOH.
a. 250.0 mL of pure w...
Chemistry: A Molecular Approach
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Propose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forwardThe 1H NMR of a compound with molecular formula C5H7BrO is shown. Determine its structure.arrow_forwardYou are provided an unknown sample with the molecular formula C8H9NO. After running the NMR, you obtain the following spectra. Name and draw the structure of the compoundarrow_forward
- The 1H NMR spectra of two compounds, each with molecular formula C11H16, are shown here. Identify the compounds.arrow_forwardHow could you distinguish the 1H NMR spectra of the following compounds?arrow_forwardDetermine the structure of the compound C6H100 whose 'H NMR spectrum is shown here. C6H100 6.5 25 7 6 3 1 Chemical shift (ppm)arrow_forward
- An NMR spectrum was run for the following compound. Assign proton NMR peaks and explain.arrow_forwardFollowing are the NMR spectra of three isomeric esters with the formula C7H14O2, all derived from propanoic acid. Provide a structure for each.arrow_forwardPropose a structure for the compound with the formula C5H11Cl that would produce the following C13 NMR spectrum.arrow_forward
- What is the structure of an unknown compound with molecular formula C6H15N that gives the following 1H NMR absorptions: 0.9 (singlet, 1 H), 1.10 (triplet, 3 H), 1.15 (singlet, 9 H), and 2.6 (quartet, 2 H) ppm?arrow_forwardPlease look at the Proton NMR spectrum and identify the proposed molecular structure. There is also a signal at 9.769 ppm indicating that an aldehyde is presentarrow_forwardEthyl benzoate PhCO₂Et has these peaks in its 13C NMR spectrum: 17.3, 61.1, 100- 150 (four peaks) and 166.8 ppm. Which peak belongs to which carbon atom? Draw the structure of ethyl benzoate and clearly identify each carbon with its corresponding chemical shift.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY