
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234722
Author: Klein
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.10, Problem 3.51P
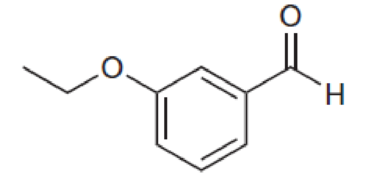
For each compound below, predict the number of signals and the location of each signal in the expected 13C NMR spectrum.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can you please confirm if this H-NMR spectrum belongs to this molecule and identify the signals of each spectrum.
Determine the multiplicity of each signal in the expected 1H and 13C NMR spectrum of the following compound.
Predict the number of signals in a proton-decoupled 13C-NMR spectrum of each compound
Chapter 3 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 3.1 - Prob. 3.2PCh. 3.1 - Prob. 3.3PCh. 3.1 - Prob. 3.4PCh. 3.1 - Prob. 3.5PCh. 3.1 - Prob. 3.6PCh. 3.1 - Prob. 3.7PCh. 3.1 - Prob. 3.8PCh. 3.1 - Prob. 3.9PCh. 3.1 - Prob. 3.10PCh. 3.1 - If you look at your answers to the previous...
Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Prob. 3.19PCh. 3.3 - Prob. 3.21PCh. 3.3 - Prob. 3.22PCh. 3.3 - Prob. 3.23PCh. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.9 - Prob. 3.43PCh. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Prob. 3.48PCh. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
a. Draw two Lewis structures for C2H6O. b. Draw three Lewis structures for C3H8O. (Hint: The two Lewis structur...
Essential Organic Chemistry (3rd Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
11.57 Draw the cis and trans isomers for each of the following: (11.6)
a. 2-pentene
b. 3-hexene
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
The equilibrium condition needs to be explained by taking example of couples dancing at a prom versus sitting a...
Living By Chemistry: First Edition Textbook
For the 3pz and 4dxz hydrogen-like atomic orbitals, sketch the following: a. The radial function R b. The radia...
Inorganic Chemistry
Saturated steam at 0.1 bar condenses with a convection coefficient of 6800W/m2K on the outside of a brass tube ...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following 1H NMR peaks were recorded on a spectrometer operating at 200 MHz. Convert each into δ units. (a) CHCl3; 1454 Hz (b) CH3Cl; 610 Hz (c) CH3OH; 693 Hz (d) CH2Cl2; 1060 Hzarrow_forwardFollowing are three compounds with the molecular formula C4H8O2 and three 1H-NMR spectra. Assign each compound its correct spectrum and assign all signals to their corresponding hydrogens. (1) (2) (3)arrow_forwardPropose structures for compounds that fit the following descriptions: (a) A hydrocarbon with seven lines in its 13C NMR spectrum (b) A six-carbon compound with only five lines in its 13C NMR spectrum (c) A four-carbon compound with three lines in its 13C NMR spectrumarrow_forward
- Predict the number of signals and the splitting pattern of each signal in the 1H-NMR spectrum of each moleculearrow_forwardWhich compound gives a signal in the 1H-NMR spectrum with a larger chemical shift, furan or cyclopentadiene? Explain.arrow_forwardThe structure of citronellal is shown below, with the carbon atoms numbered. Also shown is a table of 13C NMR signals for the molecule and the 13C-NMR DEPT spectrum. Assign each signal in the 13C-NMR spectrum to a numbered carbon in the molecule.arrow_forward
- Determine the multiplicity and predict the chemical shifts of each signal in the expected 1H NMR spectrum of the following compound.arrow_forwardhow to determine the multiplicity of each signal in the expected 1H NMR spectrumarrow_forward06) Answer the following questions for each of the substances: a) How many signals appear in the 13C NMR spectrum? b) Which signal is at the lowest frequency?arrow_forward
- How many different signals would you see in the carbon NMR of the compound below? Assume you can see them all.arrow_forwardThe 1H-NMR spectrum of 1-chloropropane shows three signals and the 1H-NMR spectrum of 2-chloropropane shows two signals. Draw these two molecules and determine the relative integrals of each signal.arrow_forward5. Determine the multiplicity and predict the chemical shifts of each signal in the expected 'H NMR spectrum of the following compound.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY