
Fundamentals of Physics, Volume 1, Chapter 1-20
10th Edition
ISBN: 9781118233764
Author: David Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 39, Problem 65P
A diatomic gas molcculc consistsof two
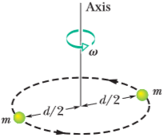
Figure 39-32 Problem 65.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem 63.
64 Martian CO, laser. Where
sunlight shines on the atmosphere
E = 0.289 eV
of Mars, carbon dioxide mole-
cules at an altitude of about 75
km undergo natural laser action.
The energy levels involved in the
action are shown in Fig. 40-26;
population inversion occurs be-
tween energy levels E, and E.
(a) What wavelength of sunlight
E = 0.165 ev
E =0
excites the molecules in the las-
Figure 40-26 Problem 64.
ing action? (b) At what wave-
length does lasing occur? (c) In what region of the electromag-
netic spectrum do the excitation and lasing wavelengths lie?
What is the transition rate for neon atoms in a He-Ne laser if the energy drop for the 632.8 nm emission is 1.96 eV and the power output is 1.0 mW.
Fo
If the wave function of a three
dimensional electronic gas is given.
√2h K
Cr24l)
L
ic
by the relationship Yer = Ae"
h
The momentum of electrons in
that gasis given by the
relationship
Chapter 39 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Ch. 39 - Prob. 1QCh. 39 - Prob. 2QCh. 39 - Prob. 3QCh. 39 - Prob. 4QCh. 39 - Prob. 5QCh. 39 - Prob. 6QCh. 39 - Prob. 7QCh. 39 - Prob. 8QCh. 39 - Prob. 9QCh. 39 - Prob. 10Q
Ch. 39 - Prob. 11QCh. 39 - Prob. 12QCh. 39 - Prob. 13QCh. 39 - Prob. 14QCh. 39 - Prob. 15QCh. 39 - Prob. 1PCh. 39 - Prob. 2PCh. 39 - Prob. 3PCh. 39 - Prob. 4PCh. 39 - Prob. 5PCh. 39 - Prob. 6PCh. 39 - Prob. 7PCh. 39 - Prob. 8PCh. 39 - Prob. 9PCh. 39 - Prob. 10PCh. 39 - Prob. 11PCh. 39 - Prob. 12PCh. 39 - Prob. 13PCh. 39 - Prob. 14PCh. 39 - Prob. 15PCh. 39 - Prob. 16PCh. 39 - Prob. 17PCh. 39 - Prob. 18PCh. 39 - Prob. 19PCh. 39 - Prob. 20PCh. 39 - Prob. 21PCh. 39 - Prob. 22PCh. 39 - Prob. 23PCh. 39 - Prob. 24PCh. 39 - Prob. 25PCh. 39 - Prob. 26PCh. 39 - Prob. 27PCh. 39 - Prob. 28PCh. 39 - Prob. 29PCh. 39 - Prob. 30PCh. 39 - Prob. 31PCh. 39 - Prob. 32PCh. 39 - Prob. 33PCh. 39 - Prob. 34PCh. 39 - Prob. 35PCh. 39 - Prob. 36PCh. 39 - Prob. 37PCh. 39 - Prob. 38PCh. 39 - Prob. 39PCh. 39 - Prob. 40PCh. 39 - Prob. 41PCh. 39 - Prob. 42PCh. 39 - Prob. 43PCh. 39 - Prob. 44PCh. 39 - Prob. 45PCh. 39 - Prob. 46PCh. 39 - Prob. 47PCh. 39 - Prob. 48PCh. 39 - Prob. 49PCh. 39 - Prob. 50PCh. 39 - Prob. 51PCh. 39 - Prob. 52PCh. 39 - Prob. 53PCh. 39 - Prob. 54PCh. 39 - Prob. 55PCh. 39 - Prob. 56PCh. 39 - Prob. 57PCh. 39 - Prob. 58PCh. 39 - Prob. 59PCh. 39 - Prob. 60PCh. 39 - Prob. 61PCh. 39 - Prob. 62PCh. 39 - Prob. 63PCh. 39 - Prob. 64PCh. 39 - A diatomic gas molcculc consistsof two atoms of...Ch. 39 - Prob. 66PCh. 39 - Prob. 67PCh. 39 - Prob. 68PCh. 39 - Prob. 69PCh. 39 - Prob. 70PCh. 39 - An old model of a hydrogen atom has the charge e...Ch. 39 - Prob. 72PCh. 39 - Prob. 73P
Additional Science Textbook Solutions
Find more solutions based on key concepts
What class of motion, natural or violent, did Aristotle attribute to motion of the Moon?
Conceptual Physics (12th Edition)
A dual-fluid heat exchanger has l0Ibm/s water entering at l00F,20psia and leaving at 50F,20psia . The other flu...
EBK FUNDAMENTALS OF THERMODYNAMICS, ENH
The temperature on the Kelvin scale.
Physics: Principles with Applications
Choose the best answer to each of the following. Explain your reasoning. What would you be most likely to find ...
The Cosmic Perspective Fundamentals (2nd Edition)
(a) Show that .
[Hint: Use integration by parts.]
(b) Let be the step function: . (1.95)
Show that .
Introduction to Electrodynamics
Instead of saying that a red-hot nail, it is correct to say it contains what?
Conceptual Integrated Science
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- At what temperature, in terms of Tc, is the critical field of a superconductor one-half its value at T = 0 K?arrow_forwardEstimate the wavelength at which plasma reflection will occur for a metal having the density of electrons N 4 x 10 m these quantities are in proper SI units : (a) 800 nm 27 -3 -30 . Take ɛo = 10- and m 105 where (b) 600 nm (c) 300 nm (d) 200 nm.arrow_forwardAn infrared spectroscopy study of a diatomic molecule XY has given the following information: We = 2377.48 cm-'; weXe 96.22 cm-1; Be 1.9982 cm- and ae 0.0190 cm-1. (a) At what frequency (expressed in wavenumbers) would the highest energy line of the P-branch of the fundamental vibrational transition appear in the IR spectrum of the molecule? (a) At what frequency (expressed in wavenumbers) would the highest energy line of the P-branch of the first overtone vibrational transition appear in the IR spectrum of the molecule?arrow_forward
- Angular momentum and Spin. An electron in an H-atom has orbital angular momentum magnitude and z-component given by L² = 1(1+1)ħ², 1 = 0,1,2,..., n-1 Lz = m₂ħ, m₁ = 0, ±1, ±2,..., ±l 3 S² = s(s+1)h² = h², 4 Consider an excited electron (n > 1) on an H-atom. Sz = msh 1 =+=ħ Show that the minimum angle that the I can have with the z-axis is given by n-1 n L.min = cos Clue: the angle a vector with magnitude V from the z-axis can be computed from cos 0 = V²/Varrow_forwardThe laboratory operation of a Laser is related to the atomic transition problem. Let E - E, = ho for two atomic states, u(@) be the radiation density, N be the number of atoms in state 1, and N2 that in state 2. B12 is the Enstein coffeicent for absorption, B21 that transition probability for emission, and A the spontaneous emission coefficient. Find N2/N1 O B12/[A+ B21] O B1zu(@)/[A + B21u(@)] O B1zu(@)/A O B1zu(@)/B21 O Au(@)/B12 Questio Not yet answered Marked out of P Flag question Forbidden transitions and selection rules suggest that O a photon has energy O a photon has mass O a photon has linear momentum O a photon has angular momentum a photon has parityarrow_forwardCompute the 2(theta) values for the first 3 x-ray diffraction peaks of a BCC metal with an atomic radius of 0.19 nm. The x-ray wavelength is 0.17 nm.arrow_forward
- An atom of iron has a radius of 156. pm and the average orbital speed of the electrons in it is about ×5.7*10^7 m/s. Calculate the least possible uncertainty in a measurement of the speed of an electron in an atom of iron. Write your answer as a percentage of the average speed, and round it to 2 significant digits.arrow_forwardIf an electron in an atom has orbital angular momentum with values limited by 3, how many values of (a) Lorb,z and (b) morb,z can the electron have? In terms of h,m, and e, what is the greatest allowed magnitude for (c) Lorb,z and (d) morb,z? (e) What is the greatest allowed magnitude for the z component of the electron’s net angular momentum (orbital plus spin)? (f) How many values (signs included) are allowed for the z component of its net angular momentum?arrow_forward(a) Using the microscopic cross sections from Hubbel et al (1975), calculate the mass attenuation coefficients for Compton scattering from the following elements: U, W, Pt, Fe and Al at the following energies: 0.1, 0.5, and 1 MeV.arrow_forward
- (4) Electronic energy level of a hydrogen atom is given by R ; п %3D 1,2, 3,... n2 E = - and R = 13.6 eV. Each energy level has degeneracy 2n2 (degeneracy is the number of equivalent configurations associated with the energy level). (a) Derive the partition function for a hydrogen atom at a constant temperature. (b) Consider that the energy level of a hydrogen atom is approximated by a two level system, n = 1,2. Estimate the mean energy at 300 K.arrow_forwardConsider 1.50 mols of a substance in thermal equilibrium at a temperature of 87.0 °C whose atoms can occupy only two energy levels separated by 5.00x102 eV, where 1 eV (electron volt) is an energy unit equal to 1.60x10 19 J. kB = 1.38x10 23 J/K , %3D NA 6.02x1023 mol1 Eg a) Find the probability of occupation of the ground state with energy E1 and the 1st excited state with energy E2? b) How many atoms in the substance (on average) are in 5.00x10 eV the ground state and in the 1st excited state? Earrow_forwardTrue or false? (a) The photon emitted in an n = 3 to n = 2 transition in the H atom has a lowerfrequency than the photon for an n = 2 to n = 1 H-atom transition. (b) The groundstate energy ofHe is about 4 times the ground-state energy of H. (c) yis zero at the nucleus for all H-atomstationary states. (d) For the ground state of the H atom, y2 is a maximum at the nucleus. (e) Themost probable value of the electron–nucleus distance in a ground-state H atom is zero. ( f ) Thesmallest allowed value of the atomic quantum number n is 0. (g) For H atom stationary stateswith l = 0, yis independent of qand f. (h) For the H-atom ground state, the electron is confinedto move on the surface of a sphere centered around the nucleus. (i) For the H-atom ground state, the electron is confined to move within a sphere of fixed radius.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax

University Physics Volume 3
Physics
ISBN:9781938168185
Author:William Moebs, Jeff Sanny
Publisher:OpenStax
Ising model | A Bird's Eye View | Solid State Physics; Author: Pretty Much Physics;https://www.youtube.com/watch?v=1CCZkHPrhzk;License: Standard YouTube License, CC-BY