
Concept explainers
Consider the substituted cyclohexane shown in the ball-and-stick model.
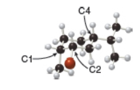
a. Label the substituents on
b. Are the substituents on
c. Are the substituents on
d. Draw the second possible conformation in the chair form, and classify it as more stable or less stable than the conformation shown in the three-dimensional model.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ALEKS 360 CHEMISTRY ACCESS
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Basic Chemistry
General, Organic, & Biological Chemistry
Chemistry: Atoms First
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Basic Chemistry (5th Edition)
- a model of cyclohexane in a chair conformation, and explain why the names “axial” and“equatorial’ are appropriate.arrow_forwarda. Draw trans-1-ethyl-2-methylcyclohexane in its lowest energy conformation. Choose a chair from the Templates toolbar at the bottom. Make sure it’s the appropriate chair, including any heteroatoms. Replace the appropriate hydrogens with the appropriate −CH3−CH3 or other groups. b. Draw the structure that corresponds with the name: isobutylcyclopentane.arrow_forwarda) Sighting down the C3-C4 bond, draw the gauche (60 degrees) and anti (180 degrees) Newman projections of 2,4-dimethylhexane. b) Circle the conformation that you drew that is lower energy.arrow_forward
- For rotation about the C3-C4 bond of 2-chlorohexane: a) Draw a Newman projection showing the most stable conformation. b) Draw a Newman projection showing the least stable conformation.arrow_forwardNeuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent.a.Label each carbon–carbon double bond as conjugated or isolated. b.Label each double bond as E or Z. c.For each conjugated system, label the given conformation as s-cis or s-trans.arrow_forwardNeuroprotectin D1 (NPD1) is synthesized in the body from highly unsaturated essential fatty acids. NPD1 is a potent natural anti-inflammatory agent. a.) Label each carbon–carbon double bond as conjugated or isolated.b.) Label each double bond as E or Z.c.) For each conjugated system, label the given conformation as s-cis or s-transarrow_forward
- Rank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −Harrow_forwardDraw the planar (Haworth) structure for cis-1-ethyl-2- methylcyclohexane. (b) Draw the two ring - flipped chair conformations. (c) Circle the more stable chair conformation (circle both if they are equal in energy). Draw the Haworth structure and the two ring-flipped conformations of 1, 2, 4- trimethylcyclohexane (Compound D) on the frames below, b) Circle the more stable chair conformation (circle both if they are equal in energy). Haworth Structurearrow_forwardOn the left is a stereorepresentation of glucose Q.Convert the stereorepresentation on the left to a chair conformation. Which substituent groups in the chair conformation are equatorial? Which are axial?arrow_forward
- a. Draw the chair 1 and 2 of Cis-1-Ethyl- 3-isopropylcyclohexane. b. Which one is more stable out of 1 or 2?arrow_forwardFocus only on carbon 1 For carbon 1, which group has the highest priority? a,b,c or d?why? What enantiomer is carbon 1? R or S?arrow_forwarda.) Draw B as a hexagon with wedges and dashed wedges to show the stereochemistry of substituents.b.) Draw a stereoisomer of A as a hexagon using wedges and dashed wedges to show the orientation of substituents.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

