
Concept explainers
Hydrocarbons like benzene are
phenols. This is an example of a general process in the body, in which an unwanted compound
(benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be
excreted more readily from the body.
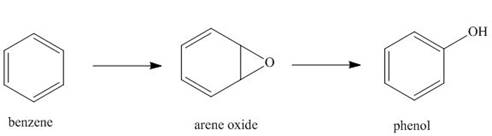
a. Classify each of these reactions as oxidation, reduction, or neither.
b. Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ALEKS 360 CHEMISTRY ACCESS
Additional Science Textbook Solutions
Organic Chemistry - Standalone book
General, Organic, & Biological Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry & Chemical Reactivity
Organic Chemistry As a Second Language: Second Semester Topics
Basic Chemistry (5th Edition)
- Draw the organic products G and H and classify/ name the chemical transformationarrow_forwardWhat changes can be made to the hydrogenation reaction to make it safer for beginner students?arrow_forwardMark the reactions in Figure 21 that will produce an alcohol as the main organic product. * A B Carrow_forward
- What reagents are used in the esterification of Alcohols and Phenols? a. Write the reaction involved in Primary Alcohol (Ethanol) and Acetyl Chloride b. Write the reaction involved in Phenol and Acetyl Chloride What is the purpose of the Chromic acid test? a. What are the reagents used? b. Write oxidation reaction of Primary Alcohols and Secondary Alcoholsarrow_forwardIndustiral Chemistry Both Tetraethyl lead and methyl-t-butyl ether have been removed from or in the process of being removed from the gasoline additive industry. Explain why each of these compounds were used so highly, and why each is considered dangerous.arrow_forwardComplete the next reactions by writing the products, reactives and experimental conditionsarrow_forward
- Draw the structure of the reactant, product(s):arrow_forwardWrite a balanced electron donor half reaction for oxidation of each following compounds: a) Benzene b) Toluene c) Ethylbenzene d) Xylenearrow_forwardComplete combustion of Propane will form what products? F (Show the equation) Which product is most useful? Garrow_forward
- Which Cyclohexanol oxidation method is “cleaner” (safer for the environment): the one using dichromate or the one using household bleach? Why (answer in terms of the waste products generated in these reactions)?arrow_forwardWhy conversion of ethanol into ethanoic acid is an oxidation reaction?arrow_forwardCould hydrochloric acid be an acceptable substitute for the acid used in dehydration reaction? explain whyarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
