
Concept explainers
For each pair of ions, determine which will have the greater number of unpaired electrons: (a) Fe2+, Fe3+; (b) P3+, P5+: (c) Cr2+, Cr3+.
(a)
Interpretation: Ground-state electronic configuration of the given set of ions has to be written.
Concept Introduction:
- Aufbau Principle tells that the orbital with the lower energy is filled with electrons first and then the filling of higher energy orbital follows. By using this valence orbital diagram can be drawn for any atoms or ions.
- Electronic configuration is the arrangement of the electrons of atoms in the orbital. For atoms and ions, the electronic configuration is written by using Pauli Exclusion Principle and Hund’s rule.
- According to Pauli Exclusion Principle, no two electrons having the same spin can occupy the same orbital.
- According to Hund’s rule, the orbital in the subshell is filled singly by one electron before the same orbital is doubly filled. When the orbital is singly filled, all the electrons have same spin. In a doubly filled orbital, there are two electrons with opposite spin.
To identify: The ion which has greater number of unpaired electrons.
Answer to Problem 4.78QP
Answer
In (a)
Explanation of Solution
Electronic configuration of
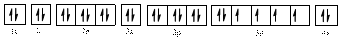
The electronic configuration of
Electronic configuration of
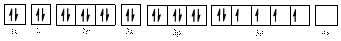
The electronic configuration of
Electronic configuration of
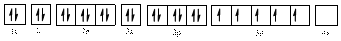
The electronic configuration of
Comparing the unpaired electrons in

By looking at the valence-orbital diagram we can see that in
(b)
Interpretation: Ground-state electronic configuration of the given set of ions has to be written.
Concept Introduction:
- Aufbau Principle tells that the orbital with the lower energy is filled with electrons first and then the filling of higher energy orbital follows. By using this valence orbital diagram can be drawn for any atoms or ions.
- Electronic configuration is the arrangement of the electrons of atoms in the orbital. For atoms and ions, the electronic configuration is written by using Pauli Exclusion Principle and Hund’s rule.
- According to Pauli Exclusion Principle, no two electrons having the same spin can occupy the same orbital.
- According to Hund’s rule, the orbital in the subshell is filled singly by one electron before the same orbital is doubly filled. When the orbital is singly filled, all the electrons have same spin. In a doubly filled orbital, there are two electrons with opposite spin.
To identify: The ion which has greater number of unpaired electrons.
Answer to Problem 4.78QP
Answer
In (b)
Explanation of Solution
Electronic configuration of
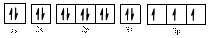
The electronic configuration of
Electronic configuration of
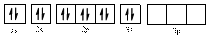
The electronic configuration of
Electronic configuration of
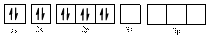
The electronic configuration of
Comparing the unpaired electrons in
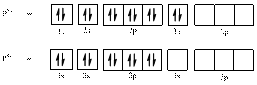
By looking at the valence-orbital diagram we can see that both electrons in
(c)
Interpretation: Ground-state electronic configuration of the given set of ions has to be written.
Concept Introduction:
- Aufbau Principle tells that the orbital with the lower energy is filled with electrons first and then the filling of higher energy orbital follows. By using this valence orbital diagram can be drawn for any atoms or ions.
- Electronic configuration is the arrangement of the electrons of atoms in the orbital. For atoms and ions, the electronic configuration is written by using Pauli Exclusion Principle and Hund’s rule.
- According to Pauli Exclusion Principle, no two electrons having the same spin can occupy the same orbital.
- According to Hund’s rule, the orbital in the subshell is filled singly by one electron before the same orbital is doubly filled. When the orbital is singly filled, all the electrons have same spin. In a doubly filled orbital, there are two electrons with opposite spin.
To identify: The ion which has greater number of unpaired electrons.
Answer to Problem 4.78QP
Answer
In (c)
Explanation of Solution
Electronic configuration of
The electronic configuration of
Electronic configuration of
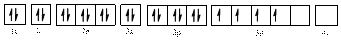
The electronic configuration of
Electronic configuration of
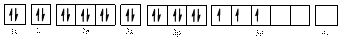
The electronic configuration of
Comparing the unpaired electrons in

By looking at the valence-orbital diagram we can see that in
Want to see more full solutions like this?
Chapter 4 Solutions
CHEM: ATOM FIRST V.2 W/ACCESS >C<
- What is the most probable position of a 1s electron in Li2+?arrow_forwardWhich transition-metal ion with a 3+ charge has the electron configuration [Kr]4d5 ?arrow_forwardWrite the ground state electron configuration and calculate Zeff for the F^2- ion. What would you expect its electron affinity to be? Explain.arrow_forward
- Write ground-state electron configurations for the following ions: K+, Mg2+, O2-, I-, Al3+,arrow_forwardWhy is the electronic configuration of the yttrium atom [Kr]4d15s2 and that of the silver atom [Kr]4d105s1?arrow_forwardWhich of the following elements would be expected to have the highest first ionization energy (IE1)?arrow_forward
- 7.137 Based on knowledge of the electronic configuration oftitanium, state which of the following compounds oftitanium is unlikely to exist: K3TiF6, K2Ti2O5, TiCl3,K2TiO4, K2TiF6.arrow_forwardThe gadolinium +3 ion (Gd3+) has advantageous magnetic properties for Magnetic Resonance Imaging (MRI) applications, specifically a high number of unpaired electrons. In fact, both neutral Gd and Gd3+ have a set of degenerate orbitals with seven unpaired electrons. a) Write the electron configuration for Gd and Gd3+ using the condensed noble gas notation. Identify which orbitals lose electrons to yield the +3 cation based on the above information. Explain your reasoning (2-3 sentences). b) Curium (Cm) is an actinide element named for Marie Curie. Cm is in the same group as Gd and also forms a +3 cation. Write the electron configuration for Cm and Cm3+. c) While +3 is the most common oxidation state for lanthanide elements, some can form +2 ions. Identify an isoelectronic ion to Gd3+ that has a +2 oxidation state and indicate which orbitals lose electrons to form that ion.arrow_forwardWhich ions in the following list are not likely to be found in chemical compounds: K2+, Cs+, Al4+, F2-, and S2+? Explain briefly.arrow_forward
- To which group of the periodic table does the general valence electron configuration ns2np4 correspond?arrow_forwardUse the electron affinity (EA) data for ClCl , BrBr , and II , along with your knowledge of periodic trends, to estimate the electron affinity of astatine to two significant figures.arrow_forwardWrite the electron configurations for (a) Ca2+, (b) Co3+, and (c) S2-.arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

