
Concept explainers
Give the IUPAC name for each compound.
a.
b.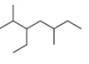
c.
d.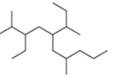
(a)
Interpretation: The IUPAC name of the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 4.8P
The IUPAC name of the given compound is
Explanation of Solution
The given compound is
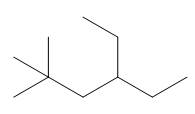
Figure 1
One should follow the given four steps to give the IUPAC name of a compound.
The first step is naming of longest parent chain.
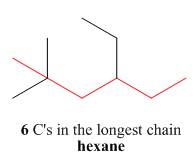
Figure 2
The second step is numbering of chain.
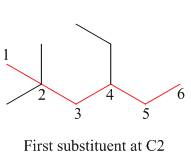
Figure 3
The third step is naming and numbering of substituents.
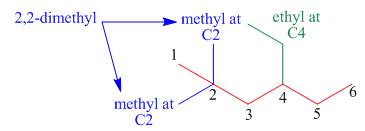
Figure 4
The fourth step is combining of all parts.
Thus, the IUPAC name of the given compound is
The IUPAC name of the given compound is
(b)
Interpretation: The IUPAC name of the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 4.8P
The IUPAC name of the given compound is
Explanation of Solution
The given compound is,
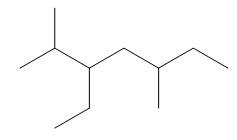
Figure 5
One should follow the given four steps to give the IUPAC name of a compound.
The first step is naming of longest parent chain.
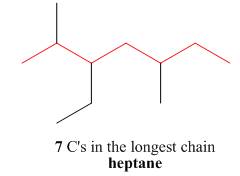
Figure 6
The second step is numbering of chain.
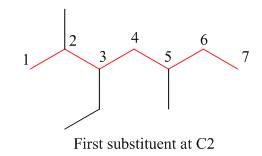
Figure 7
The third step is naming and numbering of substituents.
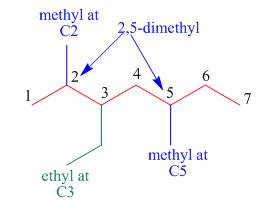
Figure 8
The fourth step is combining of all parts.
Thus, the IUPAC name of the given compound is
The IUPAC name of the given compound is
(c)
Interpretation: The IUPAC name of the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 4.8P
The IUPAC name of the given compound is
Explanation of Solution
The given compound is
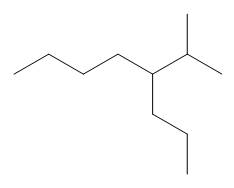
Figure 9
One should follow the given four steps to give the IUPAC name of a compound.
The first step is naming of longest parent chain.
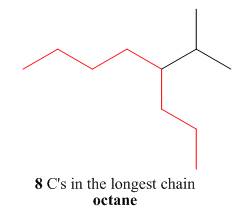
Figure 10
The second step is numbering of chain.
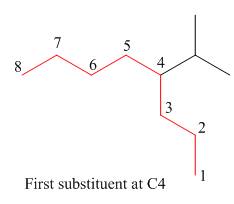
Figure 11
The third step is naming and numbering of substituents.
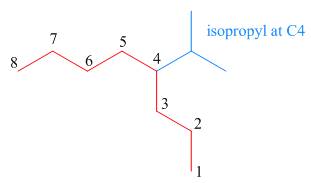
Figure 12
The fourth step is combining of all parts.
Thus, the IUPAC name of the given compound is
The IUPAC name of the given compound is
(d)
Interpretation: The IUPAC name of the given compound is to be stated.
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from structural formula are:
1. First identify the longest carbon chain.
2. The next step is to identify the groups attached to the longest chain.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
4. Use prefix di, tri, tetra if same type of substituent is present.
5. Name the substituents in alphabetical order.
Answer to Problem 4.8P
The IUPAC name of the given compound is
Explanation of Solution
The given compound is,
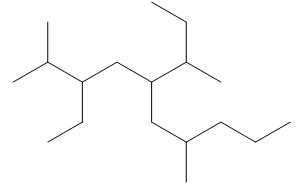
Figure 13
One should follow the given four steps to give the IUPAC name of a compound.
The first step is naming of longest parent chain.
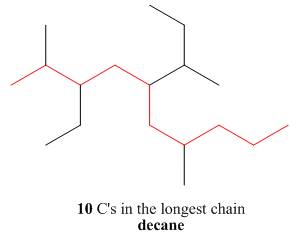
Figure 14
The second step is numbering of chain.
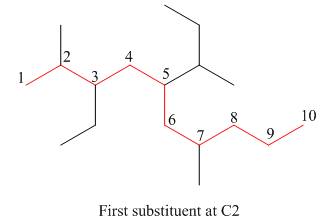
Figure 15
The third step is naming and numbering of substituents.
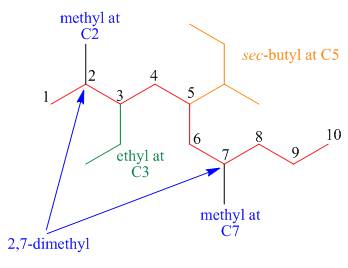
Figure 16
The fourth step is combining of all parts.
Thus, the IUPAC name of the given compound is
The IUPAC name of the given compound is
Want to see more full solutions like this?
Chapter 4 Solutions
ORGANIC CHEMISTRY W/CONNECT PKG
Additional Science Textbook Solutions
Principles of Chemistry: A Molecular Approach (3rd Edition)
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
General, Organic, and Biological Chemistry (3rd Edition)
General Chemistry: Atoms First
Organic Chemistry - Standalone book
Introduction to Chemistry
- Isooctane is the common name of the isomer of C8H18 used as the standard of 100 for the gasoline octane rating: (a) What is the IUPAC name for the compound? (b) Name the other isomers that contain a five-carbon chain with three methyl substituents.arrow_forwardALCOHOLS 1. WHY IS ETHANOL MORE SOLUBLE IN WATER THAN 1-HEXANOL? 2. WHAT IS DENATURED ALCOHOL? AND WHY IS ALCOHOL DENATURED? ETHER 1. WHY DOES DIETHYL ETHER HAVE MUCH LOWER BOILING POINT THAN 1-BUTANOL?arrow_forwardGive the IUPAC name for each alkene.arrow_forward
- Draw the structure of a molecule that fi ts each description: a. a 2 ° alcohol of molecular formula C 6H 14O b. an ether with molecular formula C 6H 14O that has a methyl group bonded to oxygen c. a 3 ° alkyl halide with molecular formula C 5H 11Brarrow_forwardDraw a cyclic ether with molecular formula C5H10Oarrow_forwardDraw an isomer of C5H10O that contains an etherarrow_forward
- What alkenes are formed when each alcohol is treated with H 2SO 4? Use the Zaitsev rule to predict the major product.arrow_forwardDraw the structure of a molecule that fi ts each description: a. a 2 ° alcohol of molecular formula C 6H 12O b. a cyclic ether with molecular formula C 5H 10O c. a 1 ° alkyl halide with molecular formula C 5H 11Clarrow_forwardWhat is the name of the alkene? CH3CH=CHCH=CH2arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


