
EBK THERMODYNAMICS: AN ENGINEERING APPR
8th Edition
ISBN: 8220100257056
Author: CENGEL
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.5, Problem 6P
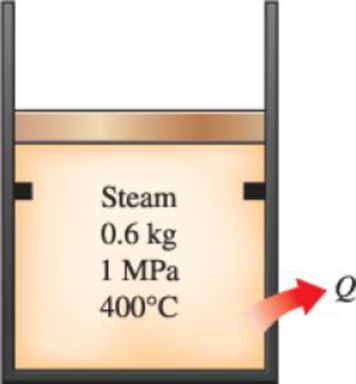
A piston–cylinder device with a set of stops initially contains 0.6 kg of steam at 1.0 MPa and 400°C. The location of the stops corresponds to 40 percent of the initial volume. Now the steam is cooled. Determine the compression work if the final state is (a) 1.0 MPa and 250°C and (b) 500 kPa. (c) Also determine the temperature at the final state in part b.
FIGURE P4–8
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A piston-cylinder device with a set of stops initially contains 0.5 kg of steam at 1.2 MPa and 500°C. The location of the stops corresponds to 30 percent of the initial volume. Now the steam is cooled. Determine the compression work if the final state is;(a) 1.2 MPa and 200°C.(b) 500 kPa(c) Also determine the temperature at the final state in part (b).
3. A piston-cylinder device with a set of stops initially contains 0.6 kg of steam at 1.0 MPa and
400°C. The location of the stops corresponds to 40 percent of the initial volume. Now the steam
is cooled. Determine the compression work if the final state is (a) 1.0 MPa and 2508C and (b)
500 kPa. (c) Also determine the temperature at the final state in part (b).
Steam
0.6 kg
1 MPa
400°C
4-65E A 10 ft³ tank contains oxygen initially at 14.7 psia and 80°F. A paddle wheel within the tank is
rotated until the pressure inside rises to 20 psia. During the process 20 Btu of heat is lost to the
surroundings. Determine the paddle-wheel work done. Neglect the energy stored in the paddle wheel.
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- 4-118 A piston-cylinder device contains helium gas initially at 100 kPa, 10°C, and 0.2 m³. The helium is now compressed in a polytropic process (PV=constant) to 700 kPa and 290°C. Determine the heat loss or gain during this process.arrow_forwarduse T(c)= 125arrow_forward4-68 An insulated piston-cylinder device initially contains 0.3 m³ of carbon dioxide at 200 kPa and 27°C. An electric switch is turned on, and a 110-V source supplies current to a resistance heater inside the cylinder for a period of 10 min. The pressure is held constant during the process, while the volume is doubled. Determine the current that passes through the resistance heater.arrow_forward
- 4-20 A piston-cylinder device contains 0.15 kg of air initially at 2 MPa and 350°C. The air is first expanded isothermally to 500 kPa, then compressed polytropically with a polytropic exponent of 1.2 to the initial pressure, and finally compressed at the constant pressure to the initial state. Determine the boundary work done for each process and the net work of the cycle.arrow_forward6- A mass of 1.2 kg of air at 150 kPa and 12 C is contained in a piston- cylinder device. The air now is compressed to a final pressure of 600 kPa. During this process, heat is transferred from the air such that the temperature inside cylinder remains constant. Calculate: (1) the work done during this process, (2) the amount of heat, and (3) show this process on P-V diagramarrow_forwardGggarrow_forward
- 3- A mass of 10 kg of saturated refrigerant-134a vapor is contained in a piston-cylinder device at 600 kPa. Now 260 kJ of heat is transferred to the refrigerant at constant pressure while a 110-V source supplies current to a resistor within the cylinder for 30 min. If the final temperature is 80°C, the electric current supplied to the resistor is: (a) 1.173A (b) 1.273A (c) 1.373A (d) 1.473A (e) 1.573Aarrow_forward(b) A 0.15 kg of air contained in a piston-cylinder device where the initial pressure was 2000 kPa and the temperature was 350°C. The substance has undergone several expansion and compression processes. The air is first expanded at a constant temperature to a pressure of 500 kPa and then compressed polytropically with a polytropic exponent of 1.2 to the initial pressure, and finally compressed isobarically to the initial state. Determine the boundary work for each process and the net work of the cycle.arrow_forwardA piston-cylinder device contains 0.5 kg of air initially at 3 MPa and 250°C.The air is first expanded at the constant pressure to 0.04 m3, thencompressed isothermally to the initial volume, and finally compressedisometrically to the initial state.1. Draw the P-V diagram.2. Determine the boundary work for each process and the net work of thecycle.3. Heat lost during the constant volume process.arrow_forward
- Q1/ A frictionless piston–cylinder device initially contains 200 L of saturated liquid refrigerant-134a. The piston is free to move, and its mass is such that it maintains a pressure of 900 kPa on the refrigerant. The refrigerant is now heated until its temperature rises to 70°C. Calculate the work done during this process. Answer: 5571 kJarrow_forwardQuestion 1: 4-20 A piston-cylinder device contains 0.15 kg of air initially at 2 MPa and 350°C. The air is first expanded isothermally to 500 kPa, then compressed polytropically with a polytropic exponent of 1.2 to the initial pressure, and finally compressed at the constant pressure to the initial state. Determine the boundary work for each process and the network of the cycle.arrow_forward4-40 Steam at 75 kPa and 8 percent quality is contained in a spring-loaded piston-cylinder device, as shown in Fig. P4-40. with an initial volume of 2 m³. Steam is now heated until its volume is 5 m³ and its pressure is 225 kPa. Determine the heat transferred to and the work produced by the steam during this process. FIGURE P4-40 www Steamarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license