
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.8, Problem 4.26YT
Scientific Practices Greenhouse Gas Spectra
- a. Use Figure 4.17 to estimate the wavelengths corresponding to the strongest IR absorbance for water vapor.
- b. Which wavelengths do you predict represent bending vibrations, and which represent stretching? Explain the basis of your predictions. Hint: Compare the IR spectrum of H2O with that of CO2.
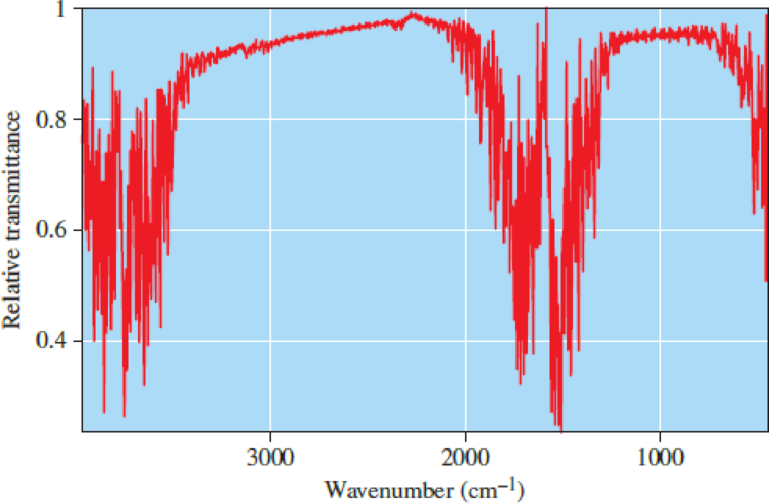
Figure 4.17
Infrared spectrum of water vapor.
Source: NIST Chemistry WebBook (http://webbook.nist.gov/chemistry)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify which of the following species have the properties listed below?
Select all that apply
?−(???????????)F−(fluorideion)
??−3(??????????)NO3−(nitrateion)
???−3(ℎ???????????????????)HCO3−(hydrogencarbonateion)
?3??4(?ℎ???ℎ????????)H3PO4(phosphoricacid)
d) can function as a buffer system component
For forensic chemistry class :
When the vehicle traveled in a curved path from right to left, which tire tracks were located on the inner portion of the set of tracks?
When the vehicle traveled in a curved path from right to left, which tires were located on the outer portion of the set of tire tracks?
Does the direction of the curved path ( right to left or left to right )change which tires create the inner and outer tire tracks? Why?
i dont need huge explanation just do all the math thankyou.
Chapter 4 Solutions
Chemistry In Context
Ch. 4.1 - You Decide What Do You Know About the Big C? In...Ch. 4.1 - Prob. 4.3YTCh. 4.1 - Prob. 4.4YTCh. 4.1 - Skill Building Polyatomic Ions II Name each of...Ch. 4.1 - Prob. 4.6YTCh. 4.2 - Prob. 4.7YTCh. 4.2 - Prob. 4.8YTCh. 4.3 - Skill Building Isotopes of Nitrogen Nitrogen (N2)...Ch. 4.3 - You Decide Marshmallows and Pennies Avogadros...Ch. 4.3 - Skill Building Calculating the Mass of Atoms...
Ch. 4.4 - Prob. 4.12YTCh. 4.4 - Prob. 4.13YTCh. 4.4 - Prob. 4.14YTCh. 4.4 - You Decide Checking Carbon from Cars A...Ch. 4.5 - Skill Building Light from the Sun Refresher In...Ch. 4.5 - Prob. 4.17YTCh. 4.6 - You Decide Evaporating Coal Mines Writing in the...Ch. 4.6 - Prob. 4.19YTCh. 4.6 - Prob. 4.20YTCh. 4.6 - Prob. 4.21YTCh. 4.7 - Skill Building MethaneFlat or Tetrahedral? a. If...Ch. 4.7 - Prob. 4.23YTCh. 4.7 - Skill Building Predicting Molecular Shapes, Part 2...Ch. 4.8 - Skill Building How Will a Molecule Stretch? For...Ch. 4.8 - Scientific Practices Greenhouse Gas Spectra a. Use...Ch. 4.8 - Scientific Practices Greenhouse Gas Spectra We...Ch. 4.8 - Prob. 4.28YTCh. 4.9 - Prob. 4.29YTCh. 4.9 - Prob. 4.30YTCh. 4.9 - Prob. 4.31YTCh. 4.9 - Prob. 4.32YTCh. 4.9 - Prob. 4.33YTCh. 4.9 - Prob. 4.34YTCh. 4.9 - Prob. 4.35YTCh. 4.9 - Prob. 4.36YTCh. 4.9 - Prob. 4.37YTCh. 4.10 - Prob. 4.38YTCh. 4.10 - Prob. 4.39YTCh. 4.10 - Prob. 4.40YTCh. 4.10 - Prob. 4.41YTCh. 4.11 - For each of the conclusions presented in Table...Ch. 4.11 - Prob. 4.43YTCh. 4.11 - Prob. 4.44YTCh. 4.12 - Skill Building Footprint Calculations Investigate...Ch. 4.12 - Scientific Practices Your Personal Share of the...Ch. 4.12 - Prob. 4.47YTCh. 4.12 - Prob. 4.48YTCh. 4.12 - Prob. 4.49YTCh. 4.12 - Prob. 4.50YTCh. 4.12 - You Decide Climate Change Insurance? Mitigation of...Ch. 4.12 - Prob. 4.52YTCh. 4.12 - You Decide The Big Question Revisited Answer these...Ch. 4 - i. Earth has increased in temperature by 6 C (10.8...Ch. 4 - The chapter concluded with a quote from John...Ch. 4 - Prob. 2QCh. 4 - Prob. 3QCh. 4 - Prob. 5QCh. 4 - a. It is estimated that 29 megajoules per square...Ch. 4 - Consider Figure 4.23. a. How does the present...Ch. 4 - Understanding Earths energy balance is essential...Ch. 4 - Explain each of these observations. a. A car...Ch. 4 - Prob. 10QCh. 4 - Draw the Lewis structure and name the molecular...Ch. 4 - Draw the Lewis structure and name the molecular...Ch. 4 - a. Draw the Lewis structure for methanol (wood...Ch. 4 - a. Draw the Lewis structure for ethene (ethylene),...Ch. 4 - Prob. 15QCh. 4 - Prob. 16QCh. 4 - Prob. 17QCh. 4 - Prob. 18QCh. 4 - Termites possess enzymes that allow them to break...Ch. 4 - Consider Figure 4.4. a. Which sector has the...Ch. 4 - Silver has an atomic number of 47. a. Give the...Ch. 4 - Silver only has two naturally occurring isotopes:...Ch. 4 - a. Calculate the average mass in grams of an...Ch. 4 - Prob. 24QCh. 4 - Prob. 25QCh. 4 - The total mass of carbon in living systems is...Ch. 4 - Prob. 27QCh. 4 - Give the number of protons, neutrons, and...Ch. 4 - Give the symbol showing the atomic number and the...Ch. 4 - Prob. 30QCh. 4 - The Arctic has been called our canary in the coal...Ch. 4 - Do you think the comment made in the cartoon is...Ch. 4 - Prob. 33QCh. 4 - Prob. 34QCh. 4 - Over the last 20 years, about 120 billion tons of...Ch. 4 - Prob. 36QCh. 4 - Prob. 37QCh. 4 - Prob. 38QCh. 4 - Ethanol, C2H5OH, can be produced from sugars and...Ch. 4 - Prob. 40QCh. 4 - Prob. 41QCh. 4 - Prob. 42QCh. 4 - Prob. 43QCh. 4 - Prob. 44QCh. 4 - Prob. 45QCh. 4 - Prob. 46QCh. 4 - Prob. 47QCh. 4 - Prob. 48QCh. 4 - Former vice president Al Gore writes in his 2006...Ch. 4 - Prob. 50QCh. 4 - Prob. 51QCh. 4 - Data taken over time reveal an increase in CO2 in...Ch. 4 - In the energy industry, 1 standard cubic foot...Ch. 4 - Prob. 54QCh. 4 - A solar oven is a low-tech, low-cost device for...Ch. 4 - In 2005, the European Union adopted a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2...
Organic Chemistry (9th Edition)
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The following are representative wavelengths in the infrared, ultraviolet, and x-ray regions of the electromagnetic spectrum, respectively: 1.0 106m, 1.0 108m, and 1.0 1010m. What is the energy of a photon of each radiation? Which has the greatest amount of energy per photon? Which has the least?arrow_forwardLike Dalton, chemists continue to model atoms using spheres. Modern models are usually drawn with a computer and use different colors to represent atoms of different elements. Which of the models below represents CO2?arrow_forwardEstimate the OH bond length in H2O from the covalent radii listed in Table 9.4.arrow_forward
- Give the orbital diagram and electron configuration for the carbon monoxide molecule, CO. What is the bond order of CO? Is the molecule diamagnetic or paramagnetic?arrow_forwardThe element cesium was discovered in 1860 by Robert Bunsen and Gustav Kirchhoff, who found two bright blue lines in the spectrum of a substance isolated from a mineral water. One of the spectral lines of cesium has a wavelength of 456 nm. What is its frequency?arrow_forwardDinitrogen difluoride (see Example 10.5) exists as cis and trans isomers. Write structural formulas for these isomers and explain (in terms of the valence bond theory of the double bond) why they exist.arrow_forward
- The C2 molecule exists in the vapor phase over carbon at high temperature. Describe the molecular orbital structure of this molecule; that is, give the orbital diagram and electron configuration. Would you expect the molecular substance to be diamagnetic or paramagnetic? What is the bond order for C2?arrow_forwardtrong>Exercise 12.3 Ozone is a very important constituent of the atmosphere. At upper levels it protects us by absorbing high-energy radiation from the sun. Near the earth's surface it produces harmful air pollution. Write the Lewis structure for ozone, O3.arrow_forwardAn atom has a line spectrum consisting of a red line and a blue line. Assume that each line corresponds to a transition between two adjacent energy levels. Sketch an energy-level diagram with three energy levels that might explain this line spectrum, indicating the transitions on this diagram. Consider the transition from the highest energy level on this diagram to the lowest energy level. How would you describe the color or region of the spectrum corresponding to this transition?arrow_forward
- The frequency of the strong red line in the spectrum of potassium is 3.91 1014/s. What is the wavelength of this light in nanometers?arrow_forwardPractice Exercise 1In which of the following species is the difference betweenthe number of protons and the number of electrons largest?(a) Ti2+ (b) P3- (c) Mn (d) Se 2- (e) Ce4+arrow_forwardQuestion 7.) What is qFe? A) -10439.1 J (B) 124725 j (C) 10439.1 j (D) -124725 jarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY