
Interpretation: The drawing showing the
Concept Introduction: When an electric field and a magnetic field are coupled together, an electromagnetic wave is formed. Here, electric and magnetic fields are perpendicular to each other as well as the direction of the wave.
Here, the wavelength of a wave is defined as the distance between two successive crests and troughs. The frequency of a wave is the number of waves passing a point in one second. The amplitude of a wave is determined by the height of the crests and troughs formed in the electromagnetic waves.
Moreover, the energy of the wave is inversely proportional to its wavelength. This can be explained by the following formula:
Here, h is Planck’s constant and c is the
Here, E is energy and
Answer to Problem 14STP
Correct answer is option (C).
Explanation of Solution
Reason for correct option:
The amplitude of the wave is the height of crests and troughs formed in the wave. From the drawings, it is clear that a wave with maximum height is given in option (C).

Thus, it is the correct option.
Reason for incorrect options:
The drawings of waves in option (A) and (B) are as follows:
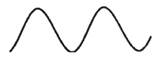
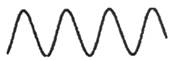
Here, the height is low as compared to the wave in option (C) thus, these waves have low amplitudes as compared to the wave in option (C).
Thus, options (A) and (B) are incorrect.
Chapter 5 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





