
Concept explainers
Write structural formulas for each of the following alcohols and
(a)
(e)
(b)
(f)
(c)
(g)
(d)
(h)
Interpretation:
The structural formulas for the given alcohols and alkyl halides are to be written.
Concept introduction:
While making the structure of an alcohol, the name of the parent alcohol is used to determine the number of atoms in the parent chain.
The substituents attached and their positions on the parent chain are determined from the name and these are drawn accordingly.
Solid lines are used to show the covalent bonds between atoms.
The structural formula of a compound shows how the atoms are arranged and bonded to each other in the compound.
Answer to Problem 19P
Solution:
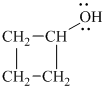
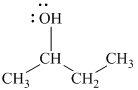

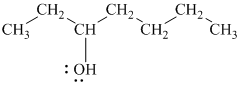
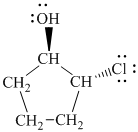
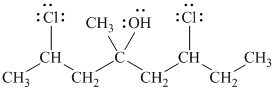
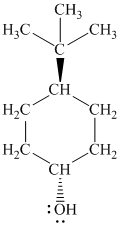
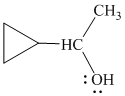
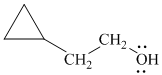
Explanation of Solution
a)
In
Therefore, the structure of
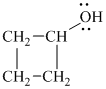
b)
In
The structure of
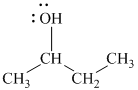
c)
In
Therefore, the structure of
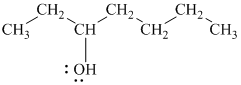
d)
In
The prefix ‘trans’ indicates that the chlorine and hydroxyl groups are on the opposite sides of the cyclopentane ring.
The structure of
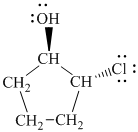
e)
In
Therefore, the structure of
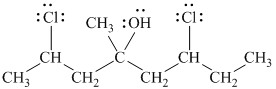
f)
In
The structure of
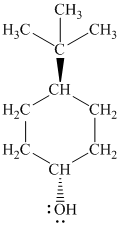
g)
In
The structure of
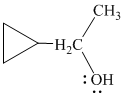
h)
In
The structure of
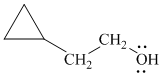
Want to see more full solutions like this?
Chapter 5 Solutions
ORGANIC CHEMISTRY-W/ACCESS >CUSTOM<
- Propose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12 c. A diene (dialkene), C5H8 d. A keto alkene, C5H8Oarrow_forwardCumene -> i. Butyl chloride ii. Dimethyl amine iii. H2, Pt Provide the bond-line structures for the major organic products obtained in each steparrow_forwardThere are several isomeric alcohols and ethers of molecular formula C5H12O. Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49 (q, 7.8 Hz, 2H), 1.85 (s, 1H) ppm Isomer B: δ = 1.19 (s, 9 H), 3.21 (s, 3H) ppmarrow_forward
- Formaldehyde, H2C=O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no carbonyl groups. Only one monobromo derivative (C3H5BrO3) of trioxane is possible. Propose a structure for trioxane.arrow_forwardAn unknown hydrocarbon Q has a formula C6H12. Q Reacts with osmium tetroxide to give a diol R. When oxidized with KMnO4 in an acidic medium, Q gives two products. One product is propanoic acid and the other a ketone S. Provide reaction equations to identify the possible structures of Q, R and S.arrow_forwardSodium triacetoxyborohydride, NaBH(OAc)3, is a mild reducing agent that reduces aldehydesmuch more quickly than ketones. It can be used to reduce aldehydes in the presence of ketones,such as in the following reaction:CH3 C CH2O OC H CH3 C CH2OCH2OHNaBH(OAc)3CH3COOH(a) Draw a complete Lewis structure for sodium triacetoxyborohydride.(b) Propose a mechanism for the reduction of an aldehyde by sodium triacetoxyborohydridearrow_forward
- 3b)Give the mechanisms for the following transformations:arrow_forward5. Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct.arrow_forwardWrite the condense structure of the following compound 1.Cyclobutanone 2. Propanenitrile 3. Pentanamidearrow_forward
- Use bond-dissociation enthalpies (Table 4-2, p. 167) to calculate values of ∆H° for the following reactions. (a) CH3¬CH3 + I2 ¡ CH3CH2I + HI(b) CH3CH2Cl + HI ¡ CH3CH2I + HCl(c) (CH3)3C¬OH + HCl ¡ (CH3)3C¬Cl + H2O (d) CH3CH2CH3 + H2 ¡ CH3CH3 + CH4 (e) CH3CH2OH + HBr ¡ CH3CH2¬Br + H2Oarrow_forwardArrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H2SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene 5,2,1,4,3 5,1,2,4,3 5,1,4,2,3 1,2,3,4,5arrow_forwardSCE // PO43- (aq) , Ag3PO4 (saturated) / Ag h a s a n I n d i c a t o r e l e c t r o d e t h a t i s a c h o o s e : l i q u i d m e m b r an e e l e ct r o d e m et a l l i c e l e c tr o d e of t he s e co n d k in d g l a ss el e c t ro d e m e t a l li c el e c t ro d e of t h e f i r st k i n darrow_forward


 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


