
Concept explainers
One Tough Bug The genus Ferroplasma consists of a few species of acid-loving archaea. One species, Ferroplasma acidarmanus, was discovered in one of the most contaminated sites in the United States: Iron Mountain Mine in California. F. acidarmamus is the main constituent of .slime streamers (a type of biofilm) growing in water draining from this abandoned copper mine (right). The water is hot (about 40°C, or 104°F), heavily laden with arsenic and other toxic metals, and has a pH of zero.
F. acidarmanus cells have an ancient energy-harvesting pathway that uses electrons pulled from iron-sulfur compounds in minerals such as pyrite. Removing electrons from these compounds dissolves the minerals, so groundwater in the mine ends up with extremely high concentrations of solutes, including metal ions such as copper, zinc, cadmium, and arsenic. The pathway also produces .sulfuric acid, which lowers the pH of the water around the cell to zero.
F. acidarmanus cells keep their internal pH at a cozy 5.0 despite Living in an environment similar to hot battery acid. However, most of the cell’s enzymes function best al much lower pH (FIGURE 5.13). Thus, researchers think F. acidarmanus may have an unknown type of internal compartment that keeps their enzymes in a highly acidic environmental.

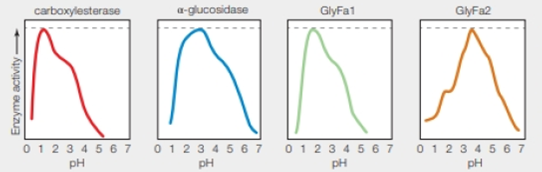
FIGURE 5.13 pH anomaly of Ferroplasma acidarmanus.
Left graphs showing pH activity profile of four enzymes isolated from Ferraplasma acidarmanus Researchers had expected all of these enzymes to function best at the calls’ cytoplasmic pH (5.0).
What do the dashed lines in the graphs signify?
To determine: What do the dashed lines in the graphs signify.
Concept introduction: Enzymes are the protein molecules that enhance the rate of the reactions without being changed by them. It acts as a biocatalyst. Each enzyme works the best within a certain range of environmental conditions that include temperature, pH, and salt concentration.
Explanation of Solution
The isolated bacteria Ferroplasma acidarmanus grow well at acidic environment (pH zero) of copper mine where the water temperature is about 40 ºC and heavily laden with arsenic and other toxic metals.
The F. acidarmanus has an ancient energy harvesting pathway. It uses the electrons from iron-sulfur compounds in the mineral such as pyrite. Removing electrons from these compounds dissolve the minerals that lead to the high concentration of metal ions such as copper, zinc, cadmium, and arsenic in groundwater. This also produces sulfuric acid which lowers the pH of the water around the cells to zero.
Despite the surrounding environment the internal pH of the bacteria is 5.0. However, most of the enzymes in bacteria functions best at much lower pH. This causes a thinking of presence of the unknown type of internal environment in the bacteria that keeps their enzymes in a highly acidic environment.
Refer to Fig. 5.13 “pH anomaly of Ferroplasma acidarmanus”, there are four different enzymes carboxylesterase, α-glucosidase, GlyFa1, and GlyFa2 which works best around pH 1.5, 3, 1.8, and 3.8 respectively. The dashed line shows the optimum activity for each enzyme.
The dashed line indicates the optimum enzyme activity for each enzyme.
Want to see more full solutions like this?
Chapter 5 Solutions
Biology: The Unity and Diversity of Life (MindTap Course List)
Additional Science Textbook Solutions
Microbiology: An Introduction (13th Edition)
Biological Science
Seeley's Anatomy & Physiology
Principles of Anatomy and Physiology
Brock Biology of Microorganisms (14th Edition)
Biology: Life on Earth with Physiology (11th Edition)
- One Tough Bug The genus Ferroplasma consists of a few species of acid-loving archaea. One species, Ferroplasma acidarmanus, was discovered in one of the most contaminated sites in the United States: Iron Mountain Mine in California. F. acidarmamus is the main constituent of .slime streamers (a type of biofilm) growing in water draining from this abandoned copper mine (right). The water is hot (about 40C, or 104F), heavily laden with arsenic and other toxic metals, and has a pH of zero. F. acidarmanus cells have an ancient energy-harvesting pathway that uses electrons pulled from iron-sulfur compounds in minerals such as pyrite. Removing electrons from these compounds dissolves the minerals, so groundwater in the mine ends up with extremely high concentrations of solutes, including metal ions such as copper, zinc, cadmium, and arsenic. The pathway also produces .sulfuric acid, which lowers the pH of the water around the cell to zero. F. acidarmanus cells keep their internal pH at a cozy 5.0 despite Living in an environment similar to hot battery acid. However, most of the cells enzymes function best al much lower pH (FIGURE 5.13). Thus, researchers think F. acidarmanus may have an unknown type of internal compartment that keeps their enzymes in a highly acidic environmental. FIGURE 5.13 pH anomaly of Ferroplasma acidarmanus. Left graphs showing pH activity profile of four enzymes isolated from Ferraplasma acidarmanus Researchers had expected all of these enzymes to function best at the calls cytoplasmic pH (5.0). What is the optimal pH for the carboxylesterase?arrow_forwardOne Tough Bug The genus Ferroplasma consists of a few species of acid-loving archaea. One species, Ferroplasma acidarmanus, was discovered in one of the most contaminated sites in the United States: Iron Mountain Mine in California. F. acidarmamus is the main constituent of .slime streamers (a type of biofilm) growing in water draining from this abandoned copper mine (right). The water is hot (about 40C, or 104F), heavily laden with arsenic and other toxic metals, and has a pH of zero. F. acidarmanus cells have an ancient energy-harvesting pathway that uses electrons pulled from iron-sulfur compounds in minerals such as pyrite. Removing electrons from these compounds dissolves the minerals, so groundwater in the mine ends up with extremely high concentrations of solutes, including metal ions such as copper, zinc, cadmium, and arsenic. The pathway also produces .sulfuric acid, which lowers the pH of the water around the cell to zero. F. acidarmanus cells keep their internal pH at a cozy 5.0 despite Living in an environment similar to hot battery acid. However, most of the cells enzymes function best al much lower pH (FIGURE 5.13). Thus, researchers think F. acidarmanus may have an unknown type of internal compartment that keeps their enzymes in a highly acidic environmental. FIGURE 5.13 pH anomaly of Ferroplasma acidarmanus. Left graphs showing pH activity profile of four enzymes isolated from Ferraplasma acidarmanus Researchers had expected all of these enzymes to function best at the calls cytoplasmic pH (5.0). Of the four enzymes profiled in the graphs, how many function optimally at a pH lower than 5? How many retain significant function at pH 5?arrow_forwardWater is important for all living organisms. The functions of water are directly related to its physical properties. Describe how the properties of water contribute of the following:( a) transpiration b. thermoregulation in the atmosphere c. pHarrow_forward
- The process of “Freeze-Drying” Foods for long-term preservation of foods that can bestored at room temperature depends upon the ability of water molecules to: a. transiently ionize to H+ and –OH ions b. form an H+ gradient when the food being frozen by oxidative phosphorylization c. acquire enough energy to break hydrogen bonds so it can escape from its neighbors and evaporate, even when the water is frozen as ice d. hydrolyze ATP into ADP + Pi e. form hydrogen bonds with oilarrow_forwardMono lake is a a soda lake (highly alkaline- pH 10) formed some 700,000 years ago. In your research, you have discovered a novel organism that is about 5x2 um in size. It is an aquatic organism, that appears to thrive in low oxygen tensions and in alkaline conditions, and appear to be chemotactic . It was found in the deeper regions of the lake with higher salinity and no oxygen. You are attempting to classify the organism as a Bacteria, Archaea or Eukarya. For each of these domains, list five (5)characteristics or structures that would allow you to classify the organism as belonging the domains. This is, for each domain, list 5 distinct characteristics or distinct structures that would place the organism in that domain. Be thoughtful, specific and detailed in your answer (by detail, I am referring to the level of detail in the text -- for example, if you believe your organism has flagella, what flagella composition/structure/movement would classify it as Archaea vs Eukarya vs…arrow_forwardA form of oxygen that has three oxygen atoms in each molecule instead of the usual two; toxic to organisms where it forms near Earth's surfacearrow_forward
- The adaptations of desert animals to their environmentinclude water conservation mechanisms. A number of theseorganisms conserve water so successfully that they neveractually drink it. They depend instead on water generatedduring metabolism. Determine how much water can beobtained by the oxidation of 1 mole of palmitic acid.arrow_forwardi just answered a hw question that i was a little confused on. i answered it but am not fully sure if it's correct. so can you take a look at check if my answer is correct and if it's not, then a explaintion would be helpful. Would you expect water molecules to move faster when there is a HIGH concentration gradient or a LOW concentration gradient? Water molecules would move faster when there is a high concentration gradient because the pressure for the molecules in the high concentration to reach equilibrium increases since there are more molecules there than in the low concentration. So as the molecules move down the concentration gradient faster to equalize the concentration on both sides due to the increased pressure, the rate of diffusion increases.arrow_forwardThe adaptation of desert animals to their environment includes water conservation mechanisms. A number of such organisms conserve water so successfully that they never drink it. They depend instead on water generated by metabolism. Determine how much water can be obtained from the complete oxidation of 1 mole of palmitic acid.arrow_forward
- Bubbles of CO2 are formed during the reaction between vinegar, water and Alka Seltzer. Provide an argument for why the CO2 molecules do not remain in the solution.arrow_forwardThe pH in your cells is decreasing. Cells have buffers to minimize changes in pH. Briefly explain what the buffer would do in this situation with respect to the H+ concentration in your cells.arrow_forwardFill in the blank. The equation describes how the reaction rate of solutes in enzymatic reactions varies with solute concentrationarrow_forward
 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning



