
Concept explainers
(a)
Interpretation:
The condensed structural formula and systematic name should be given for the molecular formula of
Concept introduction:
A condensed structural formula is a system of writing organic structures in a line of text.
Isomer: A molecule having the same molecular formula but with different chemical structure is called isomer.
Constitutional Isomers: A molecule having same molecular formula with different structural formulas (Difference in the connectivity of the molecule is called constitutional isomer).
Any organic molecule can be named by using certain rules given by IUPAC (International Union for Pure and applied chemistry).IUPAC name consists of three parts, namely Prefix, suffix and root word.
Prefix- Represents the substituent present in the molecule and its position in the root name.
Suffix- Denotes the presence of functional group if any in the molecule. It can be an alkene,
Root word - Represents the longest continuous carbon skeleton of the organic molecule.
Alkenes are a class of hydrocarbons. The carbon-carbon double bond is called as alkenes and it is also called as olefins.
(b)
Interpretation:
The E and Z isomer of the alkene has to be identified.
Concept introduction:
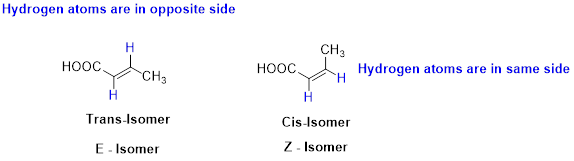
E and Z isomerism:
The two similar groups (or higher priority groups) are in same side in double bond of alkenes is called as cis isomer (or Z-isomer). Two similar groups (or higher priority groups) are opposite side in double bond of alkenes is called as trans isomer (or E-isomer).
Example:
(c)
Interpretation:
The most stable alkene has to be identified.
Concept Introduction:
Stability of alkene:
Stability of alkene depends on the following factors,
The smallest heat hydrogenation of alkene is more stable. The number of hydrogens bonded to its
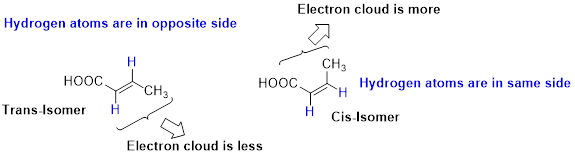
Stability of Cis–trans alkene:
The two similar groups (or higher priority groups) are in same side in double bond of alkenes is called as cis isomer (or Z-isomer). Two similar groups (or higher priority groups) are opposite side in double bond of alkenes is called as trans isomer (or E-isomer).
In cis alkene, molecule are close to each other, hence electron clouds interfere each other therefore strain in the molecule so cis alkene is less stable. Whereas molecule is away from each other, hence electron clouds are will not interfere each other therefore less strain in the molecule, so trans alkene is stable.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Essential Organic Chemistry (3rd Edition)
- Explain why unbranched alkenes can form geometric isomers while unbranched alkanes cannot. Does this explanation involve the macroscopic domain or the microscopic domain?arrow_forwardExplain why alkenes are much more reactive than alkanes towards chlorine (CI2) or bromine (Br2) in the dark at room temperature, and why alkanes do not react with HCI (g) or HBr (g) whereas alkenes do.arrow_forwardDraw 10 structural isomers of C5H10O that contain a ring.arrow_forward
- Give 5 differences of Symmetrical alkenes and Unsymmetrical alkenes .arrow_forwardHow many different alkenes can be formed from the haloalkane in Figure 20? Take stereoisomerism into account.arrow_forwardWhich alkenes show cis,trans isomerism? For each alkene that does, draw the trans isomer. Q.) 2-Pentenearrow_forward
- What is an isomer having the formula C6H12 ?Without using 2-hexenearrow_forwardName and draw structural formulas for all alkenes with the molecular formula C5H10. As you draw these alkenes, remember that cis and trans isomers are different compounds and must be counted separatelyarrow_forwardDistinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax



