
Concept explainers
(a)
Sketch thespectral content of the rectangular pulse.
(a)
Answer to Problem 34P
The spectral content of the rectangular pulse is
Explanation of Solution
Write the expression for the spectral content of the rectangular pulse,
Here,
Rewrite the above equation,
A sketch of
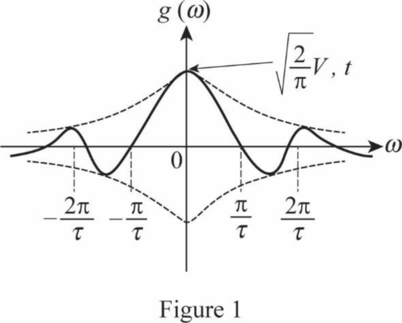
Conclusion:
In this case,
Rewrite the above equation,
Therefore, the spectral content of the rectangular pulse is
(b)
Show that a reciprocity relation
(b)
Answer to Problem 34P
A reciprocity relation
Explanation of Solution
Write the expression for the change in frequency in this case,
Here,
Write the expression for the change in time,
Here,
Conclusion:
Multiply (II) and (III),
Therefore, a reciprocity relation
(c)
The required range of frequencies to compose a pulse of width
(c)
Answer to Problem 34P
The required range of frequencies to compose a pulse of width
Explanation of Solution
Write the expression for the change in frequency in this case,
Conclusion:
For
Substitute
For
Substitute
Therefore, the required range of frequencies to compose a pulse of width
Want to see more full solutions like this?
Chapter 5 Solutions
EBK MODERN PHYSICS
- Assuming that the smallest measurable wavelength in an experiment is 0.650 fm , what is the maximum mass of an object traveling at 815 m⋅s−1 for which the de Broglie wavelength is observable?arrow_forwardA portable radio transmitter has to operate at a temperature from -5°C to 35 °C. If its signal is derived from a crystal oscillator with a temperature coefficient of +1ppm/°C and it transmits at exactly 146 MHz at 20°C, find the transmitting frequency at the lower extreme of the operating temperature range. Express the final answer in 3 decimal places and box the final answerarrow_forwardA tungsten filament of a typical incandescent lightbulb operates at a temperature near 3000 K. At what wavelength is the intensity at its maximum?arrow_forward
- The Sun is approximately an ideal blackbody radiator with a surface temperature of 5800 K. (a) Find the wavelength at which its spectral radiancy is maximum and (b) identify the type of electromagnetic wave corresponding to that wavelength. (c) As we shall discuss in Chapter 44, the universe is approximately an ideal blackbody radiator with radiation emitted when atoms first formed.Today the spectral radiancy of that radiation peaks at a wavelength of 1.06 mm (in the microwave region).What is the corresponding temperature of the universe?arrow_forwardA photon-powered spacecraft of mass 10.0 kg emits radiation of wavelength 225 nm with a power of 1.50 kW entirely in the backward direction. To what speed will it have accelerated after 10.0 y if released into free space?arrow_forwardIf the wavelength of an electron is 5.00 x 10-7 m, how fast is it moving? (b) If the electron has a speed equal to 1.00 x 107 m/s, what is its wavelength?arrow_forward
- A satellite in Earth orbit maintains a panel of solar cells of area 2.60 m2 perpendicular to the direction of the Sun’s light rays. The intensity of the light at the panel is 1.39 kW/m2. (a) At what rate does solar energy arrive at the panel? (b) At what rate are solar photons absorbed by the panel? Assume that the solar radiation is monochromatic, with a wavelength of 550 nm, and that all the solar radiation striking the panel is absorbed. (c) How long would it take for a “mole of photons” to be absorbed by the panel?arrow_forwardConsider the following statements about electromagnetic radiation and decide whether they are true or false. If they are false, correct them. (a) The total intensity of radiation emitted from a black body at absolute temperature T is directly proportional to the temperature. (b) As the temperature of a black body increases, the wavelength at which the maximum intensity is found decreases. (c) Photons of radio-frequency radiation are higher in energy than photons of ultraviolet radiation. (d) Photons of ultraviolet radiation have less energy than photons of infrared radiation. (e) The kinetic energy of an electron ejected from a metal surface when the metal is irradiated with ultraviolet radiation is independent of the frequency of the radiation. (f) The energy of a photon is inversely proportional to the wavelength of the radiation.arrow_forwardFor the thermal radiation from an ideal blackbody radiator with a surface temperature of 2000 K, let Ic represent the intensity per unit wavelength according to the classical expression for the spectral radiancy and IP represent the corresponding intensity per unit wavelength according to the Planck expression.What is the ratio Ic/IP for a wavelength of (a) 400 nm (at the blue end of the visible spectrum) and (b) 200 mm (in the far infrared)? (c) Does the classical expression agree with the Planck expression in the shorter wavelength range or the longer wavelength range?arrow_forward
- What are the energy and momentum of a photon of red light of wavelength 620 nanometers (nm)? What is the wavelength (in nm) of photons of energy 2.40 eV?arrow_forwardIf the maxwell’s wheel were to rotate twice as fast, how much would its kinetic energy increase?arrow_forwardThe surface temperature of Sun is about 6000 K. If we consider the Sun as a black body, what is the radiation power per unit of area?(Given the Stefan-Boltzmann constant is )arrow_forward
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning




