
(a)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
The systematic naming of organic compound is given by
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
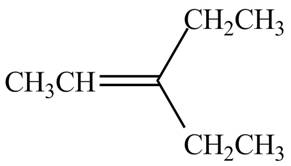
The synthesis of the given compound is shown below.
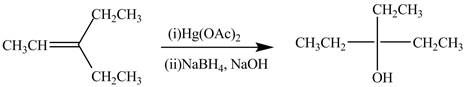
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 1
The alkene
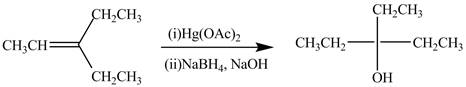
Figure 2
The preparation of the given compound from the alkene and other reagents is shown in Figure 2.
(b)
Interpretation:
The preparation of the given compound from the alkene with and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
![]()
The synthesis of the given compound is shown below.
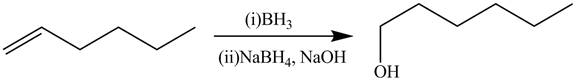
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
![]()
Figure 3
The alkene
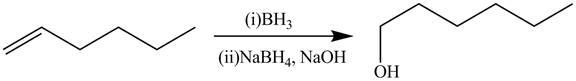
Figure 4
The preparation of the given compound from the alkene and other reagents is shown in Figure 4.
(c)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
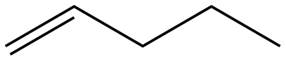
The synthesis of the given compound is shown below.
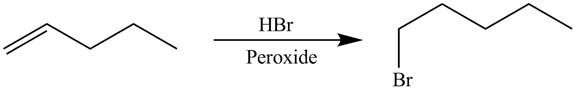
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 5
The alkene

Figure 6
The preparation of the given compound from the alkene and other reagents is shown in Figure 6.
(d)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
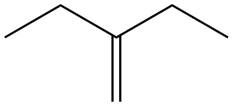
The synthesis of the given compound is shown below.
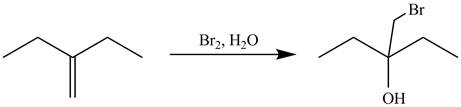
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
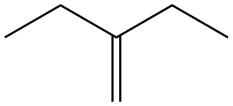
Figure 7
The alkene
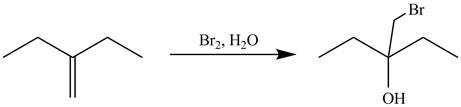
Figure 8
The preparation of the given compound from the alkene and other reagents is shown in Figure 8.
(e)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
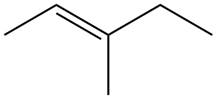
The synthesis of the given compound is shown below.
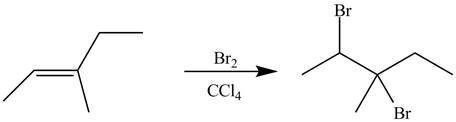
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 9
The alkene
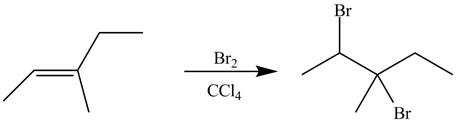
Figure 10
The preparation of the given compound from the alkene and other reagents is shown in Figure 10.
(f)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
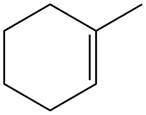
The synthesis of the given compound is shown below.
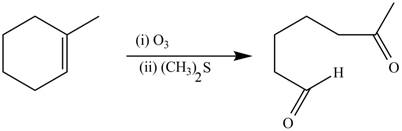
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
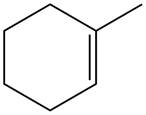
Figure 11
The alkene

Figure 12
The preparation of the given compound from the alkene and other reagents is shown in Figure 12.
(g)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
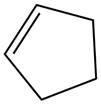
The synthesis of the given compound is shown below.
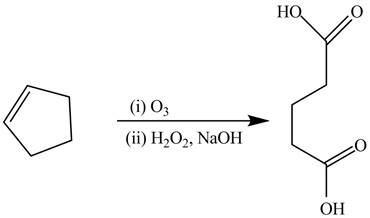
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 13
The alkene cyclopentene undergoes ozonolysis reaction followed by oxidation reaction in presence of
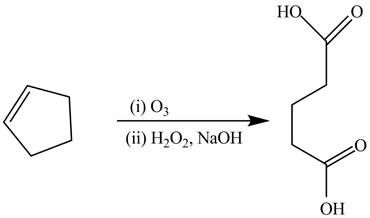
Figure 14
The preparation of the given compound from the alkene and other reagents is shown in Figure 14.
(h)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
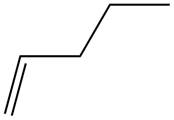
The synthesis of the given compound is shown below.
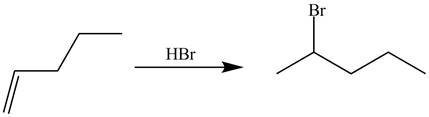
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.
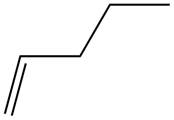
Figure 15
The alkene

Figure 16
The preparation of the given compound from the alkene and other reagentsis shown in Figure 16.
(i)
Interpretation:
The preparation of the given compound from the alkene and other reagents is to be stated.
Concept introduction:
Alkenes are the unsaturated class of organic compounds which have a double bond in their structure. The general formula of alkene is written as
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Answer to Problem 5.32AP
The alkene which is used to synthesize the given compound is shown below.
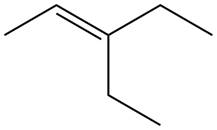
The synthesis of the given compound is shown below.
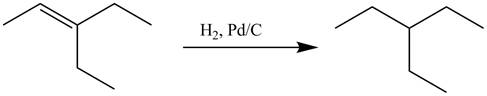
Explanation of Solution
The alkene which is used to synthesize the given compound is shown below.

Figure 17
The alkene
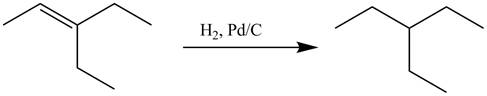
Figure 18
The preparation of the given compound from the alkene and other reagents is shown in Figure 18.
Want to see more full solutions like this?
Chapter 5 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Compound A (C5H8) readily reacts with bromine (Br2) at room temperature to discharge the purple colour of bromine and form Compound B (C5H8Br2). When Compound A is treated with H2 in the presence of a transition metal catalyst, it is converted to compound C (C5H10). When treated with HCl, compound A is converted to compound D (C5H9Cl). B, Cand D are saturated compounds. Given this information, propose structural formulas for compounds A, B, C, and D.arrow_forwardAn organic compound A of unknown structure was found to have a molecular formula C8H16. When A was poured in water and heated, compound B having a molecular formula C8H18O was formed. B upon heating with sulfuric acid was converted to C as the major product which is identical to A. Ozonolysis of C gave one molecule each of two different products D and E, both having a molecular formula C4H8O. Write the reactions involved and determine the structure of A,B,C,D and E.arrow_forwardA compound having the molecular formula C22H32O2 was isolated in 66% yield in the following reaction. Suggest a reasonable structure for this compound. What other organic compound is formed in this reaction?arrow_forward
- Compound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONSarrow_forwardCompound A was oxidized with periodic acid to give B, which after acid hydrolysis gave C. Bromine oxidation of C gave D. Suggest structural formulas, including stereochemistry, for compounds B, C, and D.arrow_forwardCompound A, C3H7Br, does not react with cold dilute potassium permanganate solution. Upon treatment with potassium hydroxide in ethanol, A gives only product B, C3H6. Unlike A, B decolourises potassium permanganate solution. Ozonolysis of Bgives C, C2H4O, and D, CH2O. Suggest the structural formulae of A, B, C and D.Write the equations for all the reactions involved.arrow_forward
- Compound F may be synthesised by the method attached: Draw the structural formulas of compounds A, C, D, E and F in the boxes providedarrow_forwardCompound P (C2H4) which is an alkene undergoes reaction with HCl to produce compound Q (C2H5Cl). Reaction of compound Q with benzene in the presence of AlCl3 as catalyst produces compound R. Then, nitration of compound R in the presence ofnH2SO4 produces two compounds, S and T. But when compound R is reacted with a hot acidified solution of alkaline KMnO4 gives compound U. Deduce the structure of compounds P to U.arrow_forwardGiven that five isomeric compounds (A, B, C, D, E) with the molecular formula C5H10O were subjected to chemical tests after undergoing Clemmensen reduction compounds A,B, and C all yielded n-pentane, the results of these tests are provided in the table below. Can you draw the structures of compounds A to E based on the outcomes of the chemical tests?arrow_forward
- An organic chemist was subjected to sodium fusion and the resulting lassaigne's extract boiled with FeSO4 and acidified with concentrated H2SO4, yielding a prussian blue color. Give the structure of one of the organic compounds which fits the description.arrow_forwardAn aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ which on heating with Br2 and KOH forms a compound ‘C’ of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B and C.arrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst to give hydrocarbon B. Hydrocarbon A also reacts with OsO4 to give the glycol C. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2COOH, and the other fragment is ketone D (R2C=O). What are the structures of A, B, C and D? Write all reactions.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





