
Interpretation:
Chiral carbon atoms present in nepetalactone has to be labelled. The number of possible stereoisomers for nepetalactone has to be given and to show that nepetalactone is a terpene.
Concept Introduction:
Terpenes are made by joining five-carbon units, usually in a head to tail-fashion.
Isoprene unit:
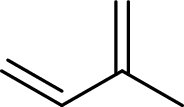
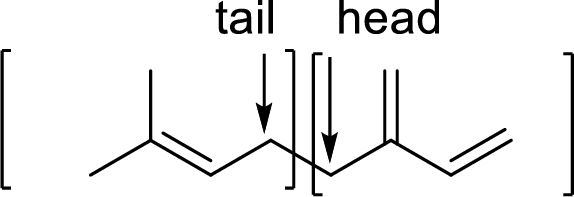
Branched end of isoprene – Head
Unbranched end of isoprene - Tail
Chirality: It refers to a Carbon atom in a molecule that contains four different substituents.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
Number of possible stereoisomers for a compound can be determined as,
Trending nowThis is a popular solution!

Chapter 5 Solutions
Organic Chemistry
- The quantitative differences in biological activity between the two enantiomers of a compound are sometimes quite large. For example, the D isomer of the drug isoproterenol, used to treat mild asthma, is 50 to 80times more effective as a bronchodilator than the L isomer. Identify the chiral center in isoproterenol. Why do the two enantiomers have such radically different bioactivity?arrow_forwardA) For a molecule with three chiral carbons the possible number of stereoisomers is: 4, 8, 6 or 2 B)Which of the following has a S configuration: (Image)arrow_forwardHow many chiral centers are in oxanamide? How many stereoisomers are possible for this compound?arrow_forward
- Pyrethrin II and pyrethrosin are two natural products isolated from plants of the chrysanthemum family. Pyrethrin II is a natural insecticide and is marketed as such. Q,) Label all chiral centers in each molecule and all carbon-carbon double bonds about which there is the possibility for cis,trans isomerism.arrow_forwardwrite structural formulas for all the compounds that are trichloro derivatives of cyclopropane, which are chiral and which are achiral?arrow_forwardA well-known non-steroidal antiinflammation drug (NSAID) exists in two stereochemical forms. Only one is biologically active. Below is the structure of the biologically active form. Identify the chiral center and determine if it is the R- or S- stereoisomerarrow_forward
- How many chirality centers are there in the molecule shown below? How many stereoisomers are possible?arrow_forwardDraw and name the six isomeric cyclopentane of molecular formula C7H14. These will include four constitutional isomers, of which two show geometric (cis-trans) stereoisomerism.arrow_forwardWhich of these do not contain a plane of symmetry?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



