
Concept explainers
Locate the stereogenic center(s) in each compound. A molecule may have zero, one, or more stereogenic centers.
a.
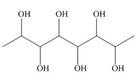 i.
i. 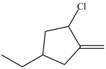
b.
c.
d.
e.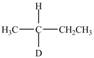 g.
g. 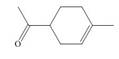 j.
j.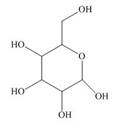
h. 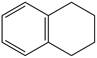
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry (Looseleaf) - With Access
- How many chirality centers exist on this molecule? What is the absolute configuration of the primary alcohol in this structure? How many lone pairs are on the structure? How many sp2 hybridized atoms are on the structure?arrow_forwardDraw all the optical isomers of 3-methylhexan-2-ol – you should be able to draw four (two pairs of enantiomers (A+B and C+D) – identify the pairs of enantiomers. Draw a circle around the 2S, 3R isomer.arrow_forwardLabel them based on the type of stereoisomerism they exhibit in. OPTICAL. NEITHER OPTICAL OR GEOMETRIC GEOMETRIC CH3CH(CBr3)CH2CH2CH3 CHCI=CHCH2CI CH3CH(CBr3)CH2CH3 CF2=CI2 CHBr=CHCH2Brarrow_forward
- 1) Are the molecules A and B... conformational isomers? Diastereomers? Enantiomers? Position isomers? Non-related? 2) What about the molecules B and C? 3) What about the molecules C and D?arrow_forwardThe facts in Section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in 2007 for the treatment of hypertension.arrow_forwardConsider the tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each six-membered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.arrow_forward
- The principles in Section 5.4A can be used to locate stereogenic centers in any molecule, no matter how complicated. Always look for carbons surrounded by four different groups. With this in mind, locate the four stereogenic centers in aliskiren, a drug introduced in 2007 for the treatment of hypertension.arrow_forwardConstruct a model in which a tetrahedral carbon atom has four different colored model atoms attached to it- red, green, orange and white representing 4 different atoms attached to the central atom. a) Does the atom have a plane of symmetry? why or why not? b) Now replace the green atom in your model with a second orange atom. Now two of the groups attached to the carbon atom are identical. Does the model now have a plane of symmetry? Describe it. c)A carbon atom has four different groups attached to the stereogenic center. Draw structural formulas for the following compound and mark stereogenic centers with as asterisk: 1-bromobutane, 2-bromobutane, 1,2-dibromobutane, 1,4-dibromobutane, 2,3-dibromobutane.arrow_forwardGlucose is a simple sugar with five substituents bonded to a six-membered ring.a. Using a chair representation, draw the most stable arrangement of these substituents on the six-membered ring.b. Convert this representation into one that uses a hexagon with wedges and dashed wedges.c. Draw a constitutional isomer of glucose.d. Draw a stereoisomer that has an axial OH group on one carbon.arrow_forward
- Only three stereoisomers are possible for 2,3-dibromo-2, 3-dichlorobutane. Draw them, indicating which pair are enantiomers (optical isomers). Why does the other isomer not have an enantiomer?arrow_forwardDraw the structure of each molecule below and put an asterisk on each tetrahedral stereocenter. i) 3,4-dichlorohexane ii) 3-bromo-1-iodo-5-methylhexane iii) 3-bromo-2,4-dimethylpentanearrow_forwardHow many chiral center(s) are present in each molecule and how many stereoisomers are possible in each case? a) 2- Chloropentane b) 3- chloropentane c) 3- Chloro- 1- pentene draw the structures of each of the molecules on paper with showing chiral centers with a star/asterisk and mention the number of the stereoisomers for each molecule.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
