
Interpretation:
The given molecular art should be identified as
Concept introduction:
Chemical reaction is process when one or more substances get converted to different products. These substances can be elements or compounds. In chemical reaction, the atoms of the reactants break down and react with the other individual elements to form new products. Through this process there is change in the identity of the substance.
Physical change is brought about by changing the physical nature of the substance without changing the composition.
Answer to Problem 5.43P
From the art pictorial representation, it is said to be a chemical change.
Explanation of Solution
Given:
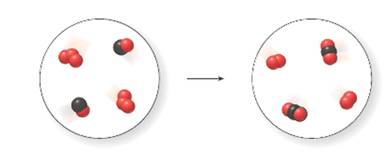
For the given molecular art, let the red balls represent oxygen atom and black balls represents carbon atoms.
Thus, in the reactant side of the molecular art, there are 3 atoms of O combine together forming ozone, O3 and one atom of O combine with one atom of C forming carbon monoxide, CO whereas in the product side of the molecular art, there are two atoms of O combined together to form dioxygen, O2 and two atoms of O combine with one atom of C forming carbon dioxide, CO2.
The equation is written as:
Since, no two substances formed at product side are similar to the initial substance on the reactant side. So, the change is chemical.
The reaction is balanced as the number of atoms of each element on the reactant side and product side are equal.
The picture depiction indicates that it is a chemical change as there is changes in the identity and alteration of the composition.
Want to see more full solutions like this?
Chapter 5 Solutions
Connect 1-Semester Access Card for General, Organic, & Biological Chemistry
- When lead(II) sulfide is heated lo high temperatures in a stream of pure oxygen gas, solid lead(II) oxide forms with the release of gaseous sulfur dioxide. Write the unbalanced chemical equation for this reaction.arrow_forwardTable 6.1 lists some clues that a chemical reaction has occurred. However, these events do not necessarily prove the existence of a chemical change. Give an example for each of the clues that is not a chemical reaction but a physical change.arrow_forwardCarbon dioxide from the atmosphere weathers, or dissolves, limestone (CaCO3) by the reaction CaCO3(s)+CO2(g)+H2O(l)Ca2(aq)+2HCO3(aq) Obtain H for this reaction. See Table 6.2 for the data.arrow_forward
- list at least three quantities that must be conserved in chemical reactions.arrow_forwardWrite a chemical equation representing the decomposition of water into two gaseous products. What are the products?arrow_forwardThe thermite reaction combines aluminum powder and iron oxide and ignites the mixture to make aluminum oxide and iron. So much energy is given off that the iron productfrequently is molten. Write a balanced chemical reaction for the thermite process and determine its H(25C).arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





