
Concept explainers
(a)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing
Answer to Problem 5.46P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of nitrogen is
The decreasing order of priority for the given groups is
(b)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.46P
The decreasing order of priority for the given groups is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
The given groups are
Therefore, the decreasing order of priority for the given groups is
The decreasing order of priority for the given groups is
(c)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.46P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of nitrogen is
Hence, the decreasing order of priority is
The decreasing order of priority for the given groups is
(d)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.46P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of carbon is
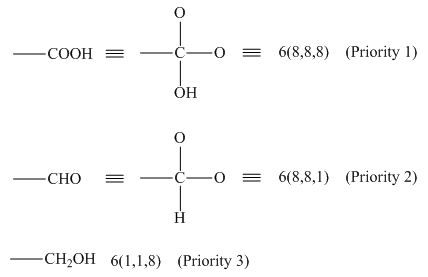
Figure 1
Hence, the decreasing order of priority is
The decreasing order of priority for the given groups is
(e)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.46P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of chlorine is
The decreasing order of priority for the given groups is
(f)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.46P
The decreasing order of priority for the given groups is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
An atom of highest atomic number gets highest priority. Therefore, in the given groups,
For an atom which is a part of multiple bonds, the priority is assigned by treating the number of multiple bonded atoms equivalent to the number of singly bonded atoms. Among,
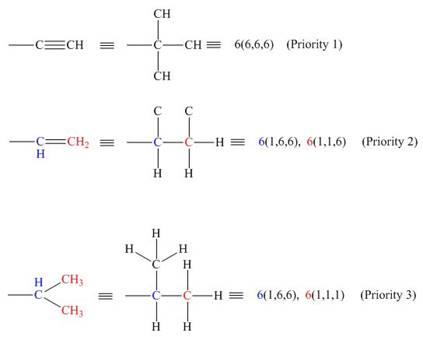
Figure 2
Hence, the decreasing order of priority for the given groups is
The decreasing order of priority for the given groups is
Want to see more full solutions like this?
Chapter 5 Solutions
ORGANIC CHEMISTRY (LOOSELEAF) >CUSTOM<
- Rank the following groups in order of decreasing priority (1 = highest, 4 = lowest)arrow_forwardArrange each set in order of decreasing priority using Cahn-Ingold-Prelog priority rules (1 for highest priority). d) _____-Cl, _____-OH, _____-H, _____-NH2 e) _____–CO2CH3, _____-CO2H, _____-OH, _____-OCH3arrow_forwardWhich group in each pair is assigned the higher priority in R,S nomenclature? a.−CD3, −CH3 b.−CH(CH3)2, −CH2OH c.−CH2Cl, −CH2CH2CH2Br d.−CH2NH2, −NHCH3arrow_forward
- Which group is of lower priority than –CH=CH2 according to Cahn-Ingold-Prelog rules. –CH=C(CH3)2 –C(CH3)3 –CH(CH3)2 –CH=CHCl a b c darrow_forwardWhich group in each pair is assigned the higher priority in R,S nomenclature? a. – CD3, – CH3 b. – CH(CH3)2, – CHOH c. – CH2Cl, – CH2CH2CH2Br d. – CH2NH2, – NHCH3arrow_forwardRank the following groups in order of decreasing priority. −NH2, −CH2NH2, −CH3, −CH2NHCH3arrow_forward
- Arrange the following group in order of increasing priority. Q) -OCH3 -CH(CH3)2 -B(CH2CH3)2 -Harrow_forwardRank the following groups in order of decreasing priority according to the Cahn-Ingold-Prelog system. I) -CH=CH2 II) -CH3 III) -CH IV) -OH 1) IV > III > I > II 2) IV > I > III > II 3) III > IV > I > II 4) II > I > III > IVarrow_forwardWhen determining R AND S rank these groups from highest to lowest priorityarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





