
Concept explainers
(a)
Interpretation:
The point on the graph corresponds to the reactants has to be given.
The given graph is,
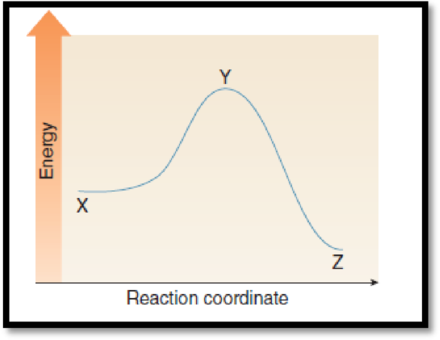
Figure 1
(b)
Interpretation:
The point on the graph corresponds to the product has to be given.
The given graph is,
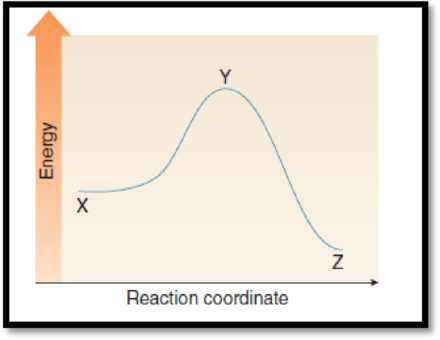
Figure 1
(c)
Interpretation:
The point on the graph corresponds to the transition state has to be given.
The given graph is,
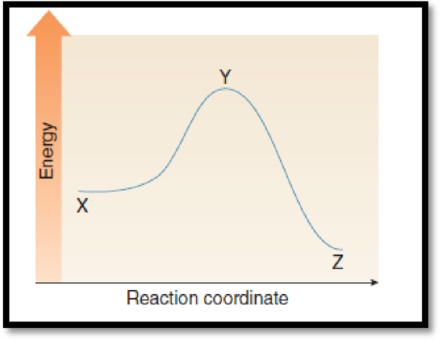
Figure 1
(d)
Interpretation:
The difference in energy between the two points which equals activation energy has to be given.
The given graph is,
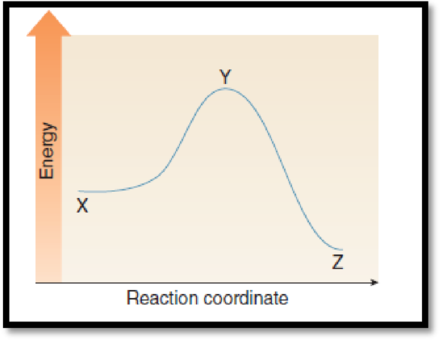
Figure 1
(e)
Interpretation:
The difference in energy between the two points which equals enthalpy has to be given.
The given graph is,
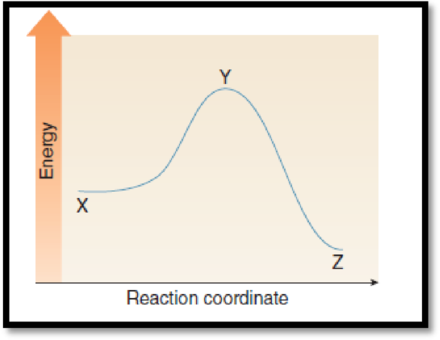
Figure 1
(f)
Interpretation:
The point with highest energy has to be given.
The given graph is,
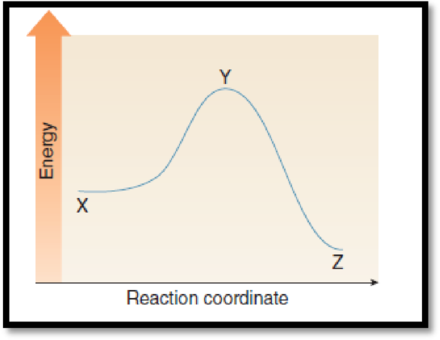
Figure 1
(g)
Interpretation:
The point with lowest energy has to be given.
The given graph is,
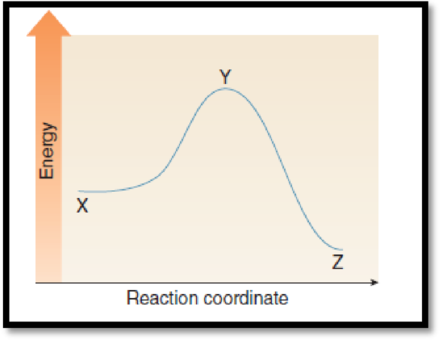
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Principles of General, Organic, Biological Chemistry
- xplain why aluminum cans make good storage containers for soft drinks. Styrofoam cups can be used to keep coffee hot and cola cold. How can this be?arrow_forward9.75 Explain why each of the following chemical equations is not a correct formation reaction. (a) 4Al(s)+3O2(g)2Al2O3(s) (b) N2(g)+32H2(g)NH3(g) (c) 2Na(s)+O(g)Na2O(s)arrow_forwardExplain why each of the following chemical equations is not a correct formation reaction: 4Al( s )+3 O 2 ( g )2 Al 2 O 3 ( s ) N 2 ( g )+ 3 2 H 2 ( g ) NH 3 ( g ) 2Na( s )+O( g ) Na 2 O( s )arrow_forward
- Draw an energy diagram graph for an endothermic reaction where no catalyst is present. Then draw an energy diagram graph for the same reaction when a catalyst is present. Indicate the similarities and differences between the two diagrams.arrow_forwardHeat was added consistently to a sample of water to producethe heating curve in Figure 15.26. Identify what is happening in Sections 1, 2, 3, and 4 on the curve.arrow_forwardConsider the following reaction 3H2(g) + CO2 (g) <=> CH3OH (g) + H2O(g) On the energy diagram, you see two energy curves: with and without catalyst. Chose correct numbers for the following: Energy of the reactant Energy of the products Overall energy of the forward reaction (ΔH) Energy of the transition state without catalyst Activation energy of the forward reaction without catalyst Activation energy of the reverse reaction with catalystarrow_forward
- Using your knowledge of temperature and reaction rate, explain why food spoils at a slower rate when refrigerated.arrow_forwardConsider the attached energy diagram for the conversion of A → G. Question: Label the overall reaction as endothermic or exothermic.arrow_forwardGiven the energy diagram below: a. The reaction releases energy. b. The reaction absorbs energy. c. The reaction occurs without a net change in energy. d. The reaction is impossible to occur.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





