
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
9th Edition
ISBN: 9780137249442
Author: Wade
Publisher: INTER PEAR
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.10A, Problem 5.16P
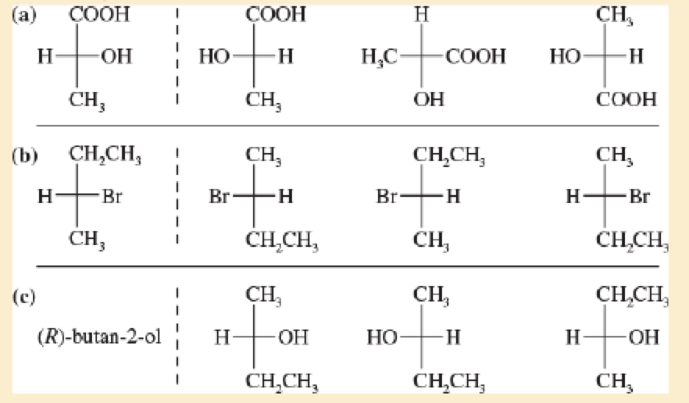
For each sot of examples, make a model of the first structure, and indicate the relationship of each of the other structures to the first structure Examples of relationships same compound, enantiomer structural isomer.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
1) Are the molecules A and B... conformational isomers? Diastereomers? Enantiomers? Position isomers? Non-related?
2) What about the molecules B and C?
3) What about the molecules C and D?
feces, gallston
3a pue
Using the structure and image below, determine the conformation of each of the designated substituents.
(Use the carbon atom numbering from the image on the left.)
HG.
HO3
bail & stick
labels
OH at C3:
H, at Cl:
Hydrogen at CB:
Submit Answer
Retry Entire Group
6 more group attempts remaining
Give typed solution not written
Chapter 5 Solutions
ORGANIC CHEMISTRY MASTERINGCHEM ACCESS
Ch. 5.2 - Determine whether the following objects are chiral...Ch. 5.2A - Prob. 5.2PCh. 5.2B - Prob. 5.3PCh. 5.2B - Prob. 5.4PCh. 5.2C - Prob. 5.5PCh. 5.3 - Prob. 5.6PCh. 5.3 - Prob. 5.7PCh. 5.4D - Prob. 5.8PCh. 5.4D - Prob. 5.9PCh. 5.4D - Prob. 5.10P
Ch. 5.5 - Prob. 5.11PCh. 5.7 - When optically pure (R)-2-bromobutane is heated...Ch. 5.7 - Prob. 5.13PCh. 5.8 - Prob. 5.14PCh. 5.9B - Draw three-dimensional representations of the...Ch. 5.10A - For each sot of examples, make a model of the...Ch. 5.10A - Draw a Fischer projection for each compound....Ch. 5.10B - Prob. 5.18PCh. 5.10C - For each Fischer projection, label each asymmetric...Ch. 5.11C - Prob. 5.20PCh. 5.13 - Prob. 5.21PCh. 5.13 - Prob. 5.22PCh. 5.15 - Prob. 5.23PCh. 5.16A - Prob. 5.24PCh. 5 - The following four structures are naturally...Ch. 5 - For each structure, 1. star () any asymmetric...Ch. 5 - Prob. 5.27SPCh. 5 - Prob. 5.28SPCh. 5 - Prob. 5.29SPCh. 5 - Prob. 5.30SPCh. 5 - Prob. 5.31SPCh. 5 - Prob. 5.32SPCh. 5 - Prob. 5.33SPCh. 5 - Prob. 5.34SPCh. 5 - For each structure, 1. draw all the stereoisomers....Ch. 5 - Prob. 5.36SPCh. 5 - Prob. 5.37SPCh. 5 - 3,4-Dimethylpent-1-ene has the formula...Ch. 5 - A graduate student was studying enzymatic...Ch. 5 - Prob. 5.40SPCh. 5 - Prob. 5.41SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
141. Design a device that uses as electrochemical cell to determine amount of
in a sample water Describe, in...
Chemistry: Structure and Properties (2nd Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Subject :- Chemistryarrow_forwardGive typing answer with explanation and conclusionarrow_forwardFor each of tthe following structural formulas, provide the indicated number of resonance structures. Use curved arrows to show the electron movement between each structure, use double headed arrow to separate the structures, and enclose all of them in a single set of bracketsarrow_forward
- Two compounds are considered as isomers if they have the…arrow_forwardClassify the pairs of molecules as not the same molecule, structural isomers, diastereomers, enantiomers or identical. Circle any molecules that are not achiral (a) (b) (c) Br... H CI H H Br H H3C Br H. CI Br H Br CI I I Br" "H H Br H CH3 Cr Brarrow_forwardIn the structure provided choose the option YES or NO to indicate if the carbon atoms are chiral. H₂N A !!! B ...!!!! OH C CIarrow_forward
- Fill in the blank. Constitutional Isomer? Conformational Isomer? Enantiomer? Diastereomer?arrow_forwardMatch the pair of compounds with the type of isomerism. Choices:a. positional isomerb. cis, trans configurationc. diastereomerd. enantiomere. functional isomerf. skeletal isomerg. E.Z configurationarrow_forward.WHICH OF THE COMPOUND SHOW GEOMETRIC ISOMERISM CH3CH=CH2 CH3C(Br)=CH(Br)CH2CH3 CH3CH2CH2CH=CHCH3 HC(CH3)=C(CH3)Cl 2-pentenearrow_forward
- What is the difference between a constitutional, confirgurational, and a conformational isomer?arrow_forwardComplex alkanes are named by using the longest continuous chain of C atoms as the basis of the name. the C atoms. 1 longest continuous chain or the parent chain of the C atoms in the formula below contains Next draw a continuous line though the C atoms in the longest continuous chain. The stem that signifies the number of C atoms in the longest continuous chain is UNDEC and the alkane name for this chain is UNDE CANE. All the groups attached to the chain of C atoms through which the line was drawn are called substituents (treat the substituent as if they are decors hang in a Christmas tree, the tree being the parent chain.) there are substituent shown in the formula. Hydrocarbon substituents are name by adding "yl" to the stem, which signifies the number of C atoms in C atoms, becomes METH YL ; "eth", which and "alk" the general stem for hydrocarbon becomes alkyl. those substituents. For example "meth" which signifies signifies 2 C atoms becomes ETHYL The names of the four substituents…arrow_forwardPls help ASAP. Pls circle the final answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License