
Organic Chemistry (8th Global Edition) - Does NOT include MasteringChemistry
8th Edition
ISBN: 9780134048147
Author: Paula Yurkanis Bruice
Publisher: Pearson Global Edition
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.12, Problem 37P
Draw a reaction coordinate diagram for the following reaction in which C is the most stable and B the least stable of the three species and the transition state going from A to B is more stable than the transition state going from B to C:
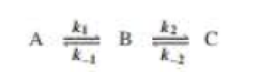
- a. How many intermediates are there?
- b. How many transition states are there?
- c. Which step has the greater rote constant in the forward direction?
- d. Which step has the greater rate constant in the reverse direction?
- e. Of the four steps, which has the greatest rate constant?
- f. Which is the rote-determining step in the forward direction?
- g. Which is the rate-determining step in the reverse direction?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a reaction coordinate diagram for the following reaction in which C is the most stable and B the least stable of the three species and the transition state going from A to B is more stable than the transition state going from B to C:a.How many intermediates are there? b. How many transition states are there? c. Which step has the greater rate constant in the forward direction? d. Which step has the greater rate constant in the reverse direction? e. Of the four steps, which has the greatest rate constant? f. Which is the rate-determining step in the forward direction? g. Which is the rate-determining step in the reverse direction?
Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, ?H°, and Ea.a. a concerted reaction with ?H° = –80 kJ/mol and Ea = 16 kJ/molb. a two-step reaction, A -> B ->C, in which the relative energy of the compounds is A < C < B, and the step A -> B is rate-determining.
Draw an energy diagram for each reaction. Label the axes, the startingmaterial, product, transition state, ΔHo, and Ea.a.) a concerted reaction with ΔHo = −80 kJ/mol and Ea = 16 kJ/molb.) a two-step reaction, A → B → C, in which the relative energy of thecompounds is A < C < B, and the step A → B is rate-determining
Chapter 5 Solutions
Organic Chemistry (8th Global Edition) - Does NOT include MasteringChemistry
Ch. 5.1 - What is the molecular formula for each of the...Ch. 5.1 - Prob. 4PCh. 5.1 - Determine the degree of unsaturation and then draw...Ch. 5.1 - Prob. 6PCh. 5.2 - What is each compounds systematic name?Ch. 5.2 - Prob. 8PCh. 5.2 - Draw the structure for each of the following: a....Ch. 5.3 - How many carbons are in the planar double-bond...Ch. 5.3 - Prob. 12PCh. 5.5 - Prob. 13P
Ch. 5.5 - Prob. 14PCh. 5.5 - Prob. 16PCh. 5.5 - Prob. 17PCh. 5.6 - a. Which of the monosubstituted cyclohexanes in...Ch. 5.6 - a. Calculate the percentage of isopropylcylohexane...Ch. 5.6 - a. for which reaction in each set will S be more...Ch. 5.6 - a. For a reaction with H = 12 kcal/ mol and S =...Ch. 5.8 - Prob. 23PCh. 5.9 - Prob. 24PCh. 5.9 - How many different alkenes can be hydrogenated to...Ch. 5.9 - The same alkane is obtained from the catalytic...Ch. 5.9 - Prob. 27PCh. 5.9 - Rank the following compounds from most stable to...Ch. 5.10 - Prob. 29PCh. 5.10 - Prob. 30PCh. 5.11 - The rate constant for a reaction can be increased...Ch. 5.11 - Prob. 33PCh. 5.11 - a. Which reaction has a greater equilibrium...Ch. 5.12 - Draw a reaction coordinate diagram for a two-step...Ch. 5.12 - a. Which step in the reaction coordinate diagram...Ch. 5.12 - Draw a reaction coordinate diagram for the...Ch. 5.13 - Prob. 38PCh. 5 - What is each compounds systematic name?Ch. 5 - Prob. 40PCh. 5 - Draw the structure of a hydrocarbon that has six...Ch. 5 - Draw the condensed structure for each of the...Ch. 5 - Prob. 43PCh. 5 - Prob. 44PCh. 5 - Prob. 45PCh. 5 - Name the following:Ch. 5 - Prob. 47PCh. 5 - Prob. 48PCh. 5 - Prob. 49PCh. 5 - In a reaction in which reactant A is in...Ch. 5 - Which bond is stronger? Briefly explain why.Ch. 5 - Prob. 52PCh. 5 - Prob. 53PCh. 5 - By following the curved red arrows, draw the...Ch. 5 - Prob. 55PCh. 5 - Prob. 56PCh. 5 - Draw structures for the following: a....Ch. 5 - Prob. 58PCh. 5 - a. Which of the following reactions has the larger...Ch. 5 - Prob. 60PCh. 5 - a. What is the equilibrium constant for a reaction...Ch. 5 - Prob. 62PCh. 5 - Prob. 63PCh. 5 - Given that the free energy of the twist-boat...Ch. 5 - Prob. 65PCh. 5 - Prob. 1PCh. 5 - Prob. 2PCh. 5 - Prob. 3PCh. 5 - Prob. 4PCh. 5 - Prob. 5PCh. 5 - Prob. 6PCh. 5 - Draw curved arrows to show the movement of the...Ch. 5 - Prob. 8PCh. 5 - Prob. 9PCh. 5 - Prob. 10P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Refer to the reaction energy diagram to answer parts (a) through (e). (a) What is the rate-limiting STEP? (Select from 1, 2, 3, or 4) (b) The TRANSITION STATE for the fastest step is: (c) In the 3rd step which INTERMEDIATE structurally resembles the transition state according to the Hammon postulate? (d) Which STEP NUMBER is endergonic? (e) The number of intermediates in the overall reaction is:arrow_forwardConsider the reaction coordinate diagram for the reaction below. How many steps is this reaction? How many intermediates are in the reaction? How many transition states are in the reaction? What is the rate limiting step?arrow_forwardConsider the attached SN2 reaction. Question: a.) Draw the structure of the transition state. b.) What is the rate equation?arrow_forward
- Consider the following two-step reaction: a.How many bonds are broken and formed in Step [1]? Would you predict the ΔHo of Step [1] to be positive or negative? b.How many bonds are broken and formed in Step [2]? Would you predict the ΔHo of Step [2] to be positive or negative? c.Which step is rate-determining? d.Draw the structure for the transition state in both steps of the mechanism. e.If ΔHooverall is negative for this two-step reaction, draw an energy diagram illustrating all of the information in parts (a)–(d).arrow_forwardDraw the free energy diagram for each of the reactions in Problem 8.33. For each diagram, include and label the overall reactants, overall products, all intermediates, and all transition states.arrow_forwardThe following is a concerted, bimolecular reaction: CH3Br + NaCN ->CH3CN + NaBr.a. What is the rate equation for this reaction?b. What happens to the rate of the reaction if [CH3Br] is doubled?c. What happens to the rate of the reaction if [NaCN] is halved?d. What happens to the rate of the reaction if [CH3Br] and [NaCN] are both increased by a factor of five?arrow_forward
- Which reaction is faster, one with Eact = +41.8 kJ/mol or one with Eact = +20.9 kJ>mol? Explain.arrow_forwardIs the structure of the transition state in the following reaction coordinate diagrams more similar to the structure of the reactant or to the structure of the product?arrow_forwardHow do I draw the separation of HI with arrows. (The reaction mechanism) Is it two half arrows or one regular arrow going to I-? Please draw it for me. Thanks!arrow_forward
- Draw an energy diagram of the following reactions. a) A one step reaction with a positive deltaG b) A one step reaction with a negative delta G C) A two-step reaction with an overall negative deltaG where the intermediates are higher energy than the reactants and the first transition state is lower in energy than the second.arrow_forwardWhich reaction is faster c or d?arrow_forwardWhich product is favored at high temp? Which product is favored at low temp? Which is never favored at any temp? Please the common intermediate of the products. Draw a reaction coordinate diagram containing all of the elements!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License