
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
7th Edition
ISBN: 9780134240152
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.4, Problem 11P
Draw and label the E and Z isomers for each of the following:
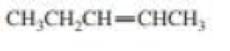

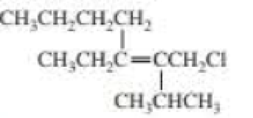
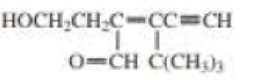
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw and label the E and Z isomers for each of the following:
Write the E and Z isomer of the following:
Choose constitutional isomers from the following compounds:
IV
Select one:
1, II, and IV
All of them
I and IV
Il and II
I, II, and II
Chapter 5 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
Ch. 5.1 - Determine the degree of unsaturation and then draw...Ch. 5.1 - Prob. 4PCh. 5.2 - Prob. 5PCh. 5.2 - Draw the structure for each of the following: a....Ch. 5.2 - What is each compounds systematic name?Ch. 5.4 - Assign relative priorities to each set of...Ch. 5.4 - Tamoxifen slows the growth of some breast tumors...Ch. 5.4 - Draw and label the E and Z isomers for each of the...Ch. 5.4 - Prob. 12PCh. 5.4 - Name each of the following:
Ch. 5.4 - Prob. 14PCh. 5.4 - Prob. 15PCh. 5.6 - Prob. 16PCh. 5.6 - Prob. 17PCh. 5.6 - Prob. 18PCh. 5.6 - Prob. 20PCh. 5.7 - a. Which of the monosubstituted cyclohexanes in...Ch. 5.7 - a. for which reaction in each set will S be more...Ch. 5.7 - a. For a reaction with H = 12 kcal/ mol and S =...Ch. 5.7 - Prob. 25PCh. 5.7 - Prob. 26PCh. 5.7 - Prob. 27PCh. 5.9 - The rate constant for a reaction can be increased...Ch. 5.9 - Prob. 30PCh. 5.9 - a. Which reaction has a greater equilibrium...Ch. 5.10 - Draw a reaction coordinate diagram for a two-step...Ch. 5.10 - a. Which step in the reaction coordinate diagram...Ch. 5.10 - Draw a reaction coordinate diagram for the...Ch. 5.11 - Prob. 35PCh. 5 - What is each compounds systematic name?Ch. 5 - Draw the structure of a hydrocarbon that has six...Ch. 5 - Draw the condensed structure for each of the...Ch. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - Name the following:Ch. 5 - Prob. 42PCh. 5 - 43. Draw the skeletal structure of...Ch. 5 - In a reaction in which reactant A is in...Ch. 5 - Which bond is stronger? Briefly explain why.Ch. 5 - Prob. 46PCh. 5 - Prob. 47PCh. 5 - Assign relative priorities to each set of...Ch. 5 - Prob. 49PCh. 5 - By following the curved red arrows, draw the...Ch. 5 - Prob. 51PCh. 5 - Draw structures for the following: a....Ch. 5 - Prob. 53PCh. 5 - a. Which of the following reactions has the larger...Ch. 5 - a. What is the equilibrium constant for a reaction...Ch. 5 - Prob. 56PCh. 5 - Prob. 57PCh. 5 - Given that the free energy of the twist-boat...Ch. 5 - Prob. 59PCh. 5 - Prob. 1PCh. 5 - Prob. 2PCh. 5 - Prob. 3PCh. 5 - Prob. 4PCh. 5 - Prob. 5PCh. 5 - Prob. 6PCh. 5 - Draw curved arrows to show the movement of the...Ch. 5 - Prob. 8PCh. 5 - Prob. 9PCh. 5 - Prob. 10P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the configuration (E) or (Z). H₂P Not a case of E vs. Z isomerism. H₂N OE OZarrow_forwardDetermine the relationship between the following pairs of compounds drawn below.Possible answers are listed in the box and can be used more than once, but each pair of structuresshould only have one answer each.1 and 2 _____3 and 4 _____5 and 6 _____7 and 8 _____A) constitutional (structural) isomersB) enantiomersC) diastereomersD) the same compoundE) unrelatedarrow_forwardDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.Both chirality and E,Z isomerismarrow_forward
- Determine the relationship between the two sets of isomers shown below. CI HO atte HO Ha OH OH CI The first set of molecules are: Diastereomers The second set of molecules are: Constitutional isomers <arrow_forwardQuestion 14 of 25 Identify the equilibrium below that shows the two chair conformations of the following compound: Br Br ^= = Type here to search i H X O O O 1 Br Br Q @ 2 W F2 # 3 E F3 A t R F4 % T F5 O A 6 F6 PrtScn & F7 7 Y U Harrow_forwardDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.Chirality but not E,Z isomerismarrow_forward
- Provide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the following compounds.arrow_forwardAmong the following pairs of isomers, identify the pair of constitutional isomers. I II III IVarrow_forwardWhich of the following molecules would be considered chiral? но OH A B Aarrow_forward
- Provide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the given compounds.arrow_forwardSelect ALL of the marked carbon atoms that are chiral: E D Br OA D OE )Farrow_forwardDraw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that shows: Q.) Neither E,Z isomerism nor chiralityarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License