
Concept explainers
(a)
Interpretation:
Check whether the given allene is chiral or achiral.
Concept Introduction:
Allenes are compounds with two or more double bonds side-by side. Such bonds are called cumulated double bonds.

Chiral centre: A chiral centre is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic centre.
An achiral carbon is a carbon having two or more identical groups around it.
Chirality: The geometric property of molecules where the structure of the molecule and its mirror image are not superimposable is known as chirality. Chiral molecules are optically active and they can rotate the plane polarized light.
Allenes shows axial chirality. Axial chirality is a stereoisomerism resulting from the non-planar arrangement of four groups in pairs about a chiral axis.
Example:
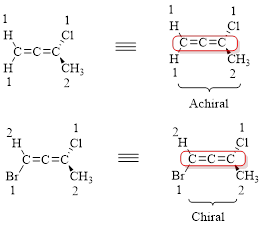
Here cumulated
(b)
Interpretation:
Check whether the given allene is chiral or achiral.
Concept Introduction:
Allenes are compounds with two or more double bonds side-by side. Such bonds are called cumulated double bonds.

Chiral centre: A chiral centre is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic centre.
An achiral carbon is a carbon having two or more identical groups around it.
Chirality: The geometric property of molecules where the structure of the molecule and its mirror image are not superimposable is known as chirality. Chiral molecules are optically active and they can rotate the plane polarized light.
Allenes shows axial chirality. Axial chirality is a stereoisomerism resulting from the non-planar arrangement of four groups in pairs about a chiral axis.
Example:

Here cumulated
(c)
Interpretation:
Check whether the given allene is chiral or achiral.
Concept Introduction:
Allenes are compounds with two or more double bonds side-by side. Such bonds are called cumulated double bonds.

Chiral centre: A chiral centre is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic centre.
An achiral carbon is a carbon having two or more identical groups around it.
Chirality: The geometric property of molecules where the structure of the molecule and its mirror image are not superimposable is known as chirality. Chiral molecules are optically active and they can rotate the plane polarized light.
Allenes shows axial chirality. Axial chirality is a stereoisomerism resulting from the non-planar arrangement of four groups in pairs about a chiral axis.
Example:

Here cumulated
(d)
Interpretation:
Check whether the given allene is chiral or achiral.
Concept Introduction:
Allenes are compounds with two or more double bonds side-by side. Such bonds are called cumulated double bonds.

Chiral centre: A chiral centre is defined as the tetrahedral carbon atom in an organic molecule that is connected to four non-identical groups/substituents. It is sometimes known as a stereo genic centre.
An achiral carbon is a carbon having two or more identical groups around it.
Chirality: The geometric property of molecules where the structure of the molecule and its mirror image are not superimposable is known as chirality. Chiral molecules are optically active and they can rotate the plane polarized light.
Allenes shows axial chirality. Axial chirality is a stereoisomerism resulting from the non-planar arrangemen6t of four groups in pairs about a chiral axis.
Example:

Here cumulated
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





