
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
9th Edition
ISBN: 9781337034623
Author: McMurry
Publisher: CENGAGE C
expand_more
expand_more
format_list_bulleted
Question
Chapter 5.SE, Problem 38AP
Interpretation Introduction
Interpretation:
The relationship between the following molecules is to be determined.
Concept introduction:
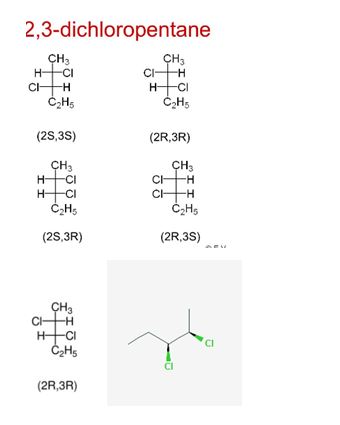
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The specific rotation of (S)-2-iodobutane is +15.90°. Determine the percentage composition of a mixture of (R)- and (S)-2-iodobutane with a specific rotation of -7.95°
If the sample of the pure R enantiomer of a molecule has a specific rotation of -40 degrees and your mixture of that molecule has an 82% ee S, what is the observed specific rotation of your sample?
1. A solution has 80% (R)-2-bromobutane and 20% (S)-2-bromobutanea. What is the “enantiomeric excess” of (R)-2-bromobutane?
b. If pure (R)-2-bromobutane rotates light 100º to the right, how much rotation would occur for a solution with 80% (R)-2-bromobutane and 20% (S)-2-bromobutane
c. If a solution has a 50/50 mixture of (R)- and (S)-2-bromobutane, what would be the enantiomeric excess and the optical purity?
2. A mixture of two enantiomers has as observed rotation of -18°. The specific rotation of the (-) enantiomers is -27°. Find the % of the two enantiomers in the solution.
Chapter 5 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
Ch. 5.2 - Prob. 1PCh. 5.2 - Prob. 2PCh. 5.2 - Prob. 3PCh. 5.2 - Prob. 4PCh. 5.3 - Is cocaine (Worked Example 5-2) dextrorotatory or...Ch. 5.3 - Prob. 6PCh. 5.5 - Prob. 7PCh. 5.5 - Prob. 8PCh. 5.5 - Prob. 9PCh. 5.5 - Assign R or S configuration to the chirality...
Ch. 5.5 - Draw a tetrahedral representation of...Ch. 5.5 - Prob. 12PCh. 5.6 - One of the following molecules (a)–(d) is...Ch. 5.6 - Prob. 14PCh. 5.6 - Assign R or S configuration to each chirality...Ch. 5.7 - Prob. 16PCh. 5.7 - Which of the following have a meso form? (Recall...Ch. 5.7 - Does the following structure represent a meso...Ch. 5.8 - Prob. 19PCh. 5.8 - Prob. 20PCh. 5.9 - Prob. 21PCh. 5.11 - Prob. 22PCh. 5.11 - Prob. 23PCh. 5.11 - The lactic acid that builds up in tired muscles is...Ch. 5.11 - The aconitase-catalyzed addition of water to...Ch. 5.SE - Which of the following structures are identical?...Ch. 5.SE - Prob. 27VCCh. 5.SE - Prob. 28VCCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 30VCCh. 5.SE - Prob. 31APCh. 5.SE - Which of the following compounds are chiral? Draw...Ch. 5.SE - Prob. 33APCh. 5.SE - Eight alcohols have the formula C5H12O. Draw them....Ch. 5.SE - Draw compounds that fit the following...Ch. 5.SE - Prob. 36APCh. 5.SE - Prob. 37APCh. 5.SE - Prob. 38APCh. 5.SE - What is the stereochemical configuration of the...Ch. 5.SE - Prob. 40APCh. 5.SE - Prob. 41APCh. 5.SE - Prob. 42APCh. 5.SE - Prob. 43APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Prob. 46APCh. 5.SE - Assign R or S configuration to each chirality...Ch. 5.SE - Assign R or S configurations to the chirality...Ch. 5.SE - Assign R or S stereochemistry to the chirality...Ch. 5.SE - Prob. 50APCh. 5.SE - Draw examples of the following: (a) A meso...Ch. 5.SE - Prob. 52APCh. 5.SE - Prob. 53APCh. 5.SE - Prob. 54APCh. 5.SE - On reaction with hydrogen gas by a platinum...Ch. 5.SE - Prob. 56APCh. 5.SE - Prob. 57APCh. 5.SE - One of the steps in fat metabolism is the...Ch. 5.SE - The dehydration of citrate to yield cis-aconitate,...Ch. 5.SE - The first step in the metabolism of glycerol,...Ch. 5.SE - One of the steps in fatty-acid biosynthesis is the...Ch. 5.SE - Prob. 62APCh. 5.SE - Draw tetrahedral representations of the two...Ch. 5.SE - The naturally occurring form of the amino acid...Ch. 5.SE - Prob. 65APCh. 5.SE - Prob. 66APCh. 5.SE - Prob. 67APCh. 5.SE - Allenes are compounds with adjacent carbon-carbon...Ch. 5.SE - Prob. 69APCh. 5.SE - Prob. 70APCh. 5.SE - How many stereoisomers of...Ch. 5.SE - Draw both cis- and trans-1,4-dimethylcyclohexane...Ch. 5.SE - Draw both cis- and trans-1,3-dimethylcyclohexane...Ch. 5.SE - cis-1,2-Dimethylcyclohexane is optically inactive...Ch. 5.SE - Prob. 75APCh. 5.SE - Prob. 76APCh. 5.SE - Prob. 77AP
Knowledge Booster
Similar questions
- Which of the following represents the correct Cahn-Ingold-Prelog priority ranking of C5H11 substituents (used to determine R/S configuration) from lowest to highest? A) CH3(CH2)4– B) (CH3)3CCH2– C) C3H7CH(CH3)– D) (CH3)2CHCH2CH2–arrow_forwardAssume that (2S,3R)-2-bromo-3-chloropentane has a specific rotation of +58°. What would be the specific rotation of the (2R,3S) isomer?arrow_forwardNaproxen, a nonsteroidal anti-inflammatory drug that is the active ingredient in Aleve (p. 115), has a specific rotation of +66. One commercial preparation results in a mixture with a 97% enantiomeric excess. a. Does naproxen have the R or the S configuration? b. What percent of each enantiomer is obtained from the commercial preparation?arrow_forward
- For bromochloromethanol that has R configuration Interchange any 2substituent, what is the absolute r/s configuration?arrow_forwardThe absolute value of the specific rotation of an enantiomer 2-butanol is 13.5o. What is the percentage of each enantiomer (R and S) in a mixture that has a specific rotation of -8.5o? The (R)-enantiomer is levorotatory. Be clear and detailed.arrow_forwardFind the optical purity of a sample that has an observed rotation of +4.5o and a specific rotation of +66o.arrow_forward
- The specific rotation of (S)-2-iodobutane is +15.90°. Predict the specific rotation of (R)-2-iodobutane.arrow_forwardDetermine whether the circled hydrogens are enantiotopic, diastereotopic, heterotopic, or homotopic.arrow_forwardDo Sn2 reactions always happen with inversion of stereochemistry, and does the stereocenter always change from R to S or S to R?arrow_forward
- Consider the reaction between (1S,3S)‑1‑chloro‑3‑methylcyclopentane and methanethiol in the presence of sodium hydroxide. (a) Draw the organic product and clearly indicate stereochemistry by showing the hydrogen on the chirality centers and using wedge and dash bonds. (b) Then analyze the stereochemistry of the product. racemic chiral achiral (1R, 3S) (1R, 3R) (1S, 3S)arrow_forwardIn some nucleophilic substitutions under SN1 conditions, completeracemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data.(b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.arrow_forwardThe specific rotation of (R)carvone is -61o. A chemist prepared a mixture of (R)-carvone and (S)-carvone, and the mixture had an observed rotation of -47o. What is the % enantiomeric excess (ee) of the mixture?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning