
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 18PS
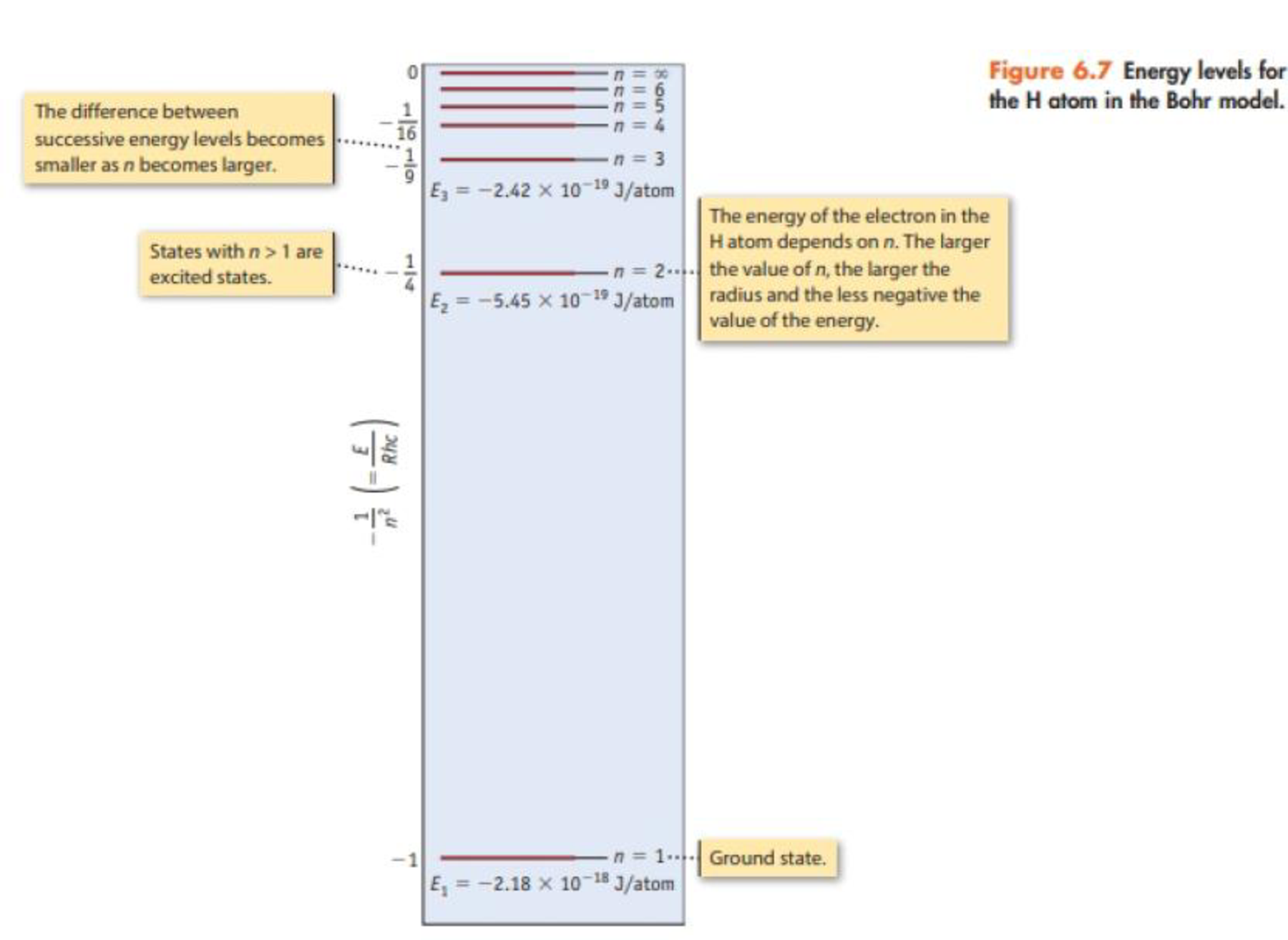
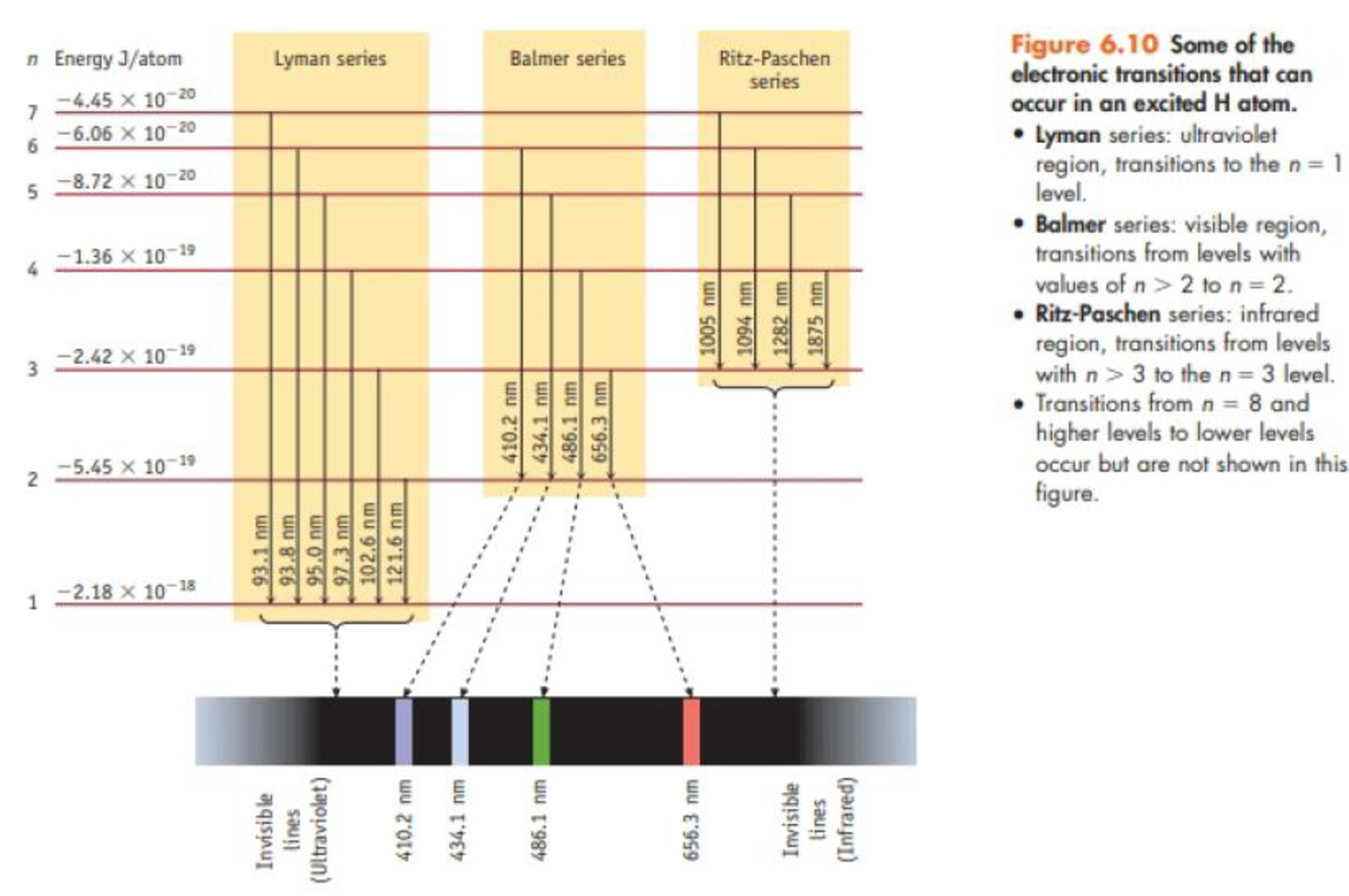
Consider only transitions involving the n = 1 through n = 4 energy levels for the hydrogen atom (see Figures 6.7 and 6.10).
- (a) How many emission lines are possible, considering only the four quantum levels?
- (b) Photons of the lowest energy are emitted in a transition from the level with n = ____to a level with n = ____
- (c) The emission line having the shortest wavelength corresponds to a transition from the level with n = ____ to the level with n = _____
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 6 Solutions
Chemistry & Chemical Reactivity
Ch. 6.1 - (a) Which color in the visible spectrum has the...Ch. 6.2 - Calculate the energy per mole of photons for the...Ch. 6.3 - Prob. 6.3CYUCh. 6.3 - The Lyman series of spectral lines for the H atom,...Ch. 6.4 - Calculate the wavelength associated with a neutron...Ch. 6.7 - Which has the longer wavelength, visible light or...Ch. 6.7 - Calculate the energy per mole of photons (in...Ch. 6.7 - Prob. 2.1ACPCh. 6.7 - Does the main emission line for SrCl2 have a...Ch. 6.7 - Prob. 2.3ACP
Ch. 6.7 - Helium absorbs light at 587.6 nm. What is the...Ch. 6.7 - Prob. 3.2ACPCh. 6.7 - Prob. 3.3ACPCh. 6.7 - Prob. 3.4ACPCh. 6.7 - Prob. 3.5ACPCh. 6 - Answer the following questions based on Figure...Ch. 6 - Consider the colors of the visible spectrum. (a)...Ch. 6 - Traffic signals are often now made of LEDs...Ch. 6 - Suppose you are standing 225 m from a radio...Ch. 6 - Green light has a wavelength of 5.0 102 nm. What...Ch. 6 - Violet light has wavelength of about 410 nm. What...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - Place the following types of radiation in order of...Ch. 6 - Place the following types of radiation in order of...Ch. 6 - An energy of 3.3 1019 J/atom is required to cause...Ch. 6 - You are an engineer designing a switch that works...Ch. 6 - The most prominent line in the spectrum of mercury...Ch. 6 - The most prominent line in the spectrum of neon is...Ch. 6 - A line in the Balmer series of emission lines of...Ch. 6 - What are the wavelength and frequency of the...Ch. 6 - Consider only transitions involving the n = 1...Ch. 6 - Consider only transitions involving the n = 1...Ch. 6 - The energy emitted when an electron moves from a...Ch. 6 - If energy is absorbed by a hydrogen atom in its...Ch. 6 - Calculate the wavelength and frequency of light...Ch. 6 - Calculate the wavelength and frequency of light...Ch. 6 - An electron moves with a velocity of 2.5 X 108...Ch. 6 - A beam of electrons (m = 9.11 X 1031 kg/electron)...Ch. 6 - Calculate the wavelength, in nanometers,...Ch. 6 - A rifle bullet (mass = 1.50 g) has a velocity of...Ch. 6 - (a) When n = 4, what are the possible values of ?...Ch. 6 - (a) When n = 4, = 2, and m = 1, to what orbital...Ch. 6 - A possible excited state of the H atom has the...Ch. 6 - A possible excited state for the H atom has an...Ch. 6 - How many subshells occur in the electron shell...Ch. 6 - Prob. 32PSCh. 6 - Explain briefly why each of the following is not a...Ch. 6 - Which of the following represent valid sets of...Ch. 6 - What is the maximum number of orbitals that can be...Ch. 6 - What is the maximum number of orbitals that can be...Ch. 6 - Explain briefly why each of the following is not a...Ch. 6 - Explain briefly why each of the following is not a...Ch. 6 - State which of the following orbitals cannot exist...Ch. 6 - State which of the following orbitals cannot exist...Ch. 6 - Write a complete set of quantum numbers (n, , m)...Ch. 6 - Write a complete set of quantum numbers (n, , and...Ch. 6 - A particular orbital has n = 4 and = 2. What must...Ch. 6 - A given orbital has a magnetic quantum number of m...Ch. 6 - Prob. 45PSCh. 6 - Prob. 46PSCh. 6 - Which of the following are applicable when...Ch. 6 - Prob. 48GQCh. 6 - Give the number of nodal surfaces through the...Ch. 6 - What is the maximum number of s orbitals found in...Ch. 6 - Match the values of l shown in the table with...Ch. 6 - Sketch a picture of the 90% boundary surface of an...Ch. 6 - Complete the following table.Ch. 6 - Excited H atoms have many emission lines. One...Ch. 6 - An advertising sign gives off red light and green...Ch. 6 - Radiation in the ultraviolet region of the...Ch. 6 - A cell phone sends signals at about 850 MHz (where...Ch. 6 - Assume your eyes receive a signal consisting of...Ch. 6 - If sufficient energy is absorbed by an atom, an...Ch. 6 - Suppose hydrogen atoms absorb energy so that...Ch. 6 - Rank the following orbitals in the H atom in order...Ch. 6 - How many orbitals correspond to each of the...Ch. 6 - Cobalt-60 is a radioactive isotope used in...Ch. 6 - Exposure to high doses of microwaves can cause...Ch. 6 - When the Sojourner spacecraft landed on Mars in...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - Answer the following questions as a summary quiz...Ch. 6 - Answer the following questions as a summary quiz...Ch. 6 - For an electron in a hydrogen atom, calculate the...Ch. 6 - A solution of KMnO4 absorbs light at 540 nm (page...Ch. 6 - Prob. 71ILCh. 6 - The spectrum shown here is for aspirin. The...Ch. 6 - The infrared spectrum for methanol. CH3OH, is...Ch. 6 - Bohr pictured the electrons of the atom as being...Ch. 6 - Light is given off by a sodium- or...Ch. 6 - Prob. 76SCQCh. 6 - What does wave-particle duality mean? What are its...Ch. 6 - Prob. 79SCQCh. 6 - Suppose you live in a different universe where a...Ch. 6 - A photon with a wavelength of 93.8 nm strikes a...Ch. 6 - Explain why you could or could not measure the...Ch. 6 - Prob. 83SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Light with a wavelength of 425 nm fell on a potassium surface, and electrons were ejected at a speed of 4.88 105 m/s. What energy was expended in removing an electron from the metal? Express the answer in joules (per electron) and in kilojoules per mole (of electrons).arrow_forwardA bright violet line occurs at 435.8 nm in the emission spectrum of mercury vapor. What amount of energy, in joules, must be released by an electron in a mercury atom to produce a photon of this light?arrow_forwardThe energy emitted when an electron moves from a higher energy state to a lower energy state in any atom can be observed as electromagnetic radiation. (a) Which involves the emission of less energy in the H atom, an electron moving from n = 4 to n = 2 or an electron moving from n = 3 to n = 2? (b) Which involves the emission of more energy in the H atom, an electron moving from n = 4 to n = 1 or an electron moving from n = 5 to n = 2? Explain fully.arrow_forward
- A particular microwave oven delivers 750 watts. (A watt is a unit of power, which is the joules of energy delivered, or used, per second.) If the oven uses microwave radiation of wavelength 12.6 cm, how many photons of this radiation are required to heat 1.00 g of water 1.00C, assuming that all of the photons are absorbed?arrow_forwardLight with a wavelength of 405 nm fell on a strontium surface, and electrons were ejected. If the speed of an ejected electron is 3.36 105 m/s, what energy was expended in removing the electron from the metal? Express the answer in joules (per electron) and in kilojoules per mole (of electrons).arrow_forward6.29 A mercury atom emits light at many wavelengths, two of which are at 435.8 and 546.1 nm. Both of these transitions are to the same final state. (a) What is the energy difference between the two states for each transition? (b) lf a transition between the two higher energy states could be observed, what would be the frequency of the light?arrow_forward
- 6.85 The visible lines in the hydrogen atom emission spectrum arise from transitions with a final state with n = 2. In what spectral region should we expect to find transitions that have a final state of n = 1 ? Explain your reasoning using an energy level diagram similar to the one in Problem 6.26.arrow_forwardWhich of the following statements is (are) true? I. The product of wavelength and frequency of light is a constant. II. As the energy of electromagnetic radiation increases, its frequency decreases. III. As the wavelength of light increases, its frequency increases. a I only b II only c III only d I and III only e II and III onlyarrow_forwardPlanck originated the idea that energies can be quantized. What does the term quantized mean? What was Planck trying to explain when he was led to the concept of quantization of energy? Give the formula he arrived at and explain each of the terms in the formula.arrow_forward
- Three emission lines involving three energy levels in an atom occur at wavelengths x, 1.5x, and 3.0x nanometers. Which wavelength corresponds to the transition from the highest to the lowest of the three energy levels?arrow_forwardThe most prominent line in the emission spectrum of aluminum is at 396.15nm. What is the frequency of this line? What is the energy of one photon with this wavelength? Of 1.00 mol of these photons?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
The Bohr Model of the atom and Atomic Emission Spectra: Atomic Structure tutorial | Crash Chemistry; Author: Crash Chemistry Academy;https://www.youtube.com/watch?v=apuWi_Fbtys;License: Standard YouTube License, CC-BY